Liver Cancer
The Common Vein Copyright 2009
Ganesh Athappan MD Ashley Davidoff MD
Definition
Liver cancer is an aggressive growth disorder initiated by a cell or a group of renegade cells, either as a primary event in the liver, but commonly as a metastatic diesease. As a primary event, the usual causes include spontaneous genetic aberration, a carcinogen in the environment, or less commonly by an inherited genetic abnormality.
The structuralchanges are characterized by proliferation and space occupation of non functioning tissue, with a variety of macroscopic morphologies, but often characterized at a cellular level by large hyperchromatic nuclii with diminished cytoplasm.
Functionally the rebel cells and cell group parasitize nutrition and oxygen, but do not contribute to the overall function of the mother organ, in this case the liver, or to the community at large.
The disease is complicatedby local spread within the organ and surrounding tissues, displacing well meaning and well functioning tissue. Continued uncontrolled growth results in the invasion of blood vessels and lymphatics and spread to distant organs where metastatic disease repeats the pattern of continued advance of rebellious parasitization on normal tissue. Other local complications include bleeding, necrosis, and obstruction. Systemic and non specific complications include fatigue, weight loss, night sweats and pain
The diagnosis is suspected clinicallywhen a patient presents with unexplained weight loss, or a new mass is felt on clinical examination.
Imagingcharacteristics include finding a mass, often multicentric, characteristically round or infltrating, that shows evidence of local invasion and or metastatic disease. When cancer is suspected, pathological confirmation is almost universally indicated to confirm the diagnosis, and to evaluate and classify the type and virulence of the cancer. Types of biopsy include aspiration technique where individual cells are sucked up into a needle, core biopsy where a cutting needle provides a small sample of tissue, and incisional biopsy where a part of a lump or a sample is removed using a scalpel.
Staging the disease is essential in the diagnostic workup since treatment plans depend on the extent and location of the disease.
Treatmentoptions depend on staging of the cancer. In the early stages the goal of treatment is cure and may include surgical resection. In advanced stages the role of treatment is control of disease and prolongation of life. This may include surgery, chemotherapy and radiation. Recent advances include localized chemical ablation with alcohol for example, or thermal or electrical ablation.
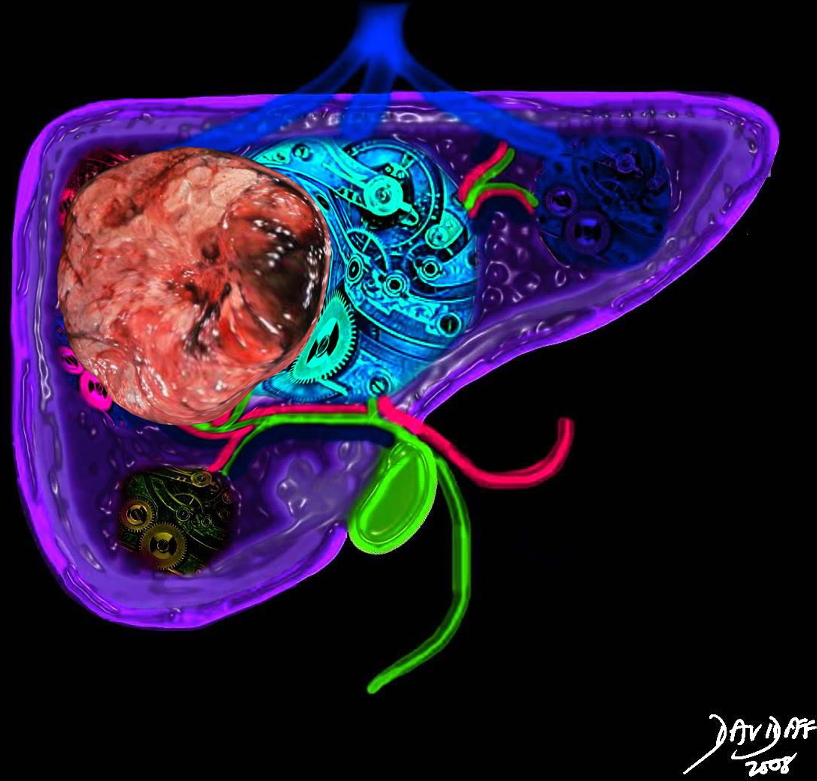 Liver Cancer Occupying Space Distorting Structure and Function Liver Cancer Occupying Space Distorting Structure and Function |
| The liver is a vital structure, and is considered the metabolic warehouse of the body. The image reflects it 24/7, clockwork nature processing churning and producing products for ongoing metabolism. The growth of any tumor in the liver will distort structure and affect function. This image reflects a large tumor throwing a “spanner” in the wheels of production.
44427b02sb02.8s liver cancer mass effect space occupation structural distortion Davidoff art copyright 2009 all rights reserved |
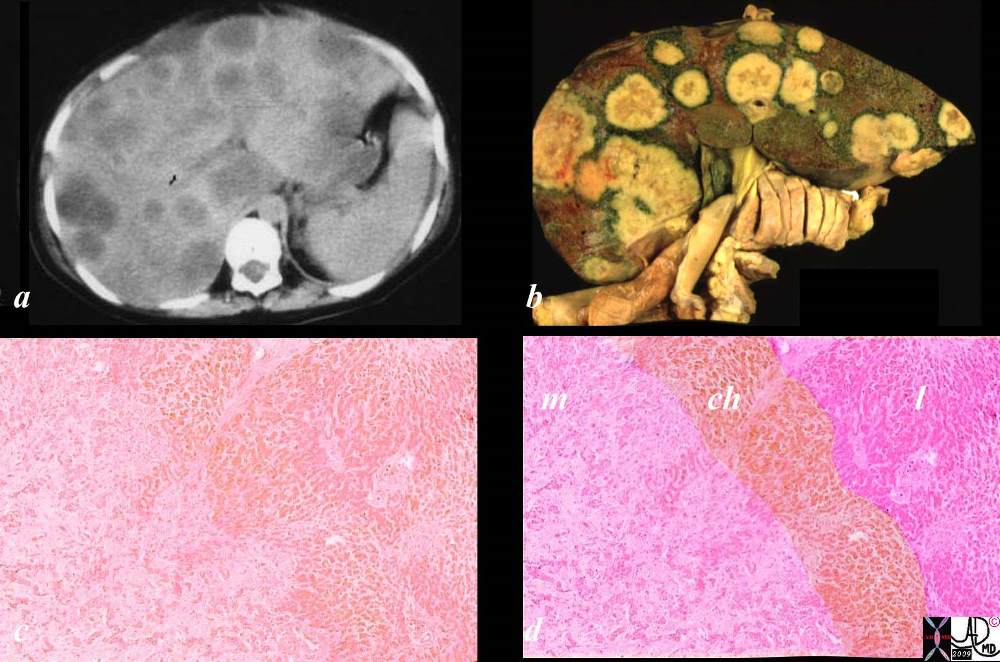
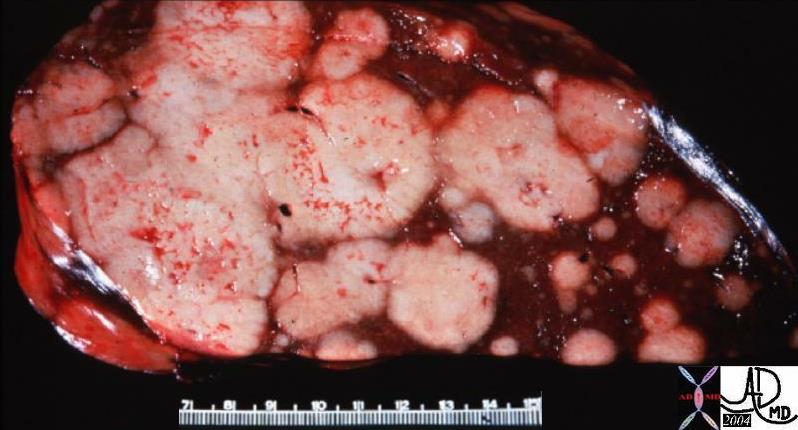 Space Occupation in the Liver Space Occupation in the Liver |
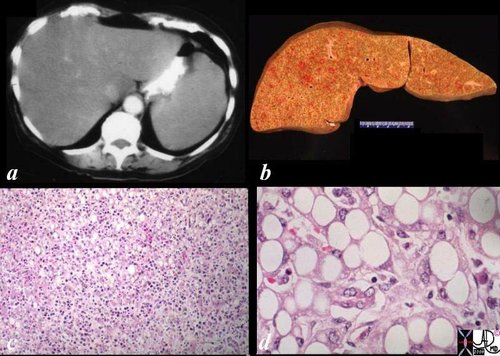
| The gross pathology specimen shows malignant tissue (white) occupying at least 80% of the normal liver parenchyma (red). The liver has remarkable tolerance for space occupation before showing ill effects, and also has remarkable regenerative capabilities. In this instance the disease was caused by metatsatic disease. Even the liver in this instance could not tolerate the degree of invasion, and the patient finally succumbed to the disease.
12362 liver fx masses dx metastatic carcinoma of the breast + grosspathology Courtesy Ashley Davidoff MD |
Hepatic cancer has varied origins including those that arise primarily from liver elements, and those are metastatic deposits.
The tumors that arise from liver elements include
hepatocellular carcinoma
cholangiocarcinoma
hepatoblasoma
liver sarcoma
malignant hemangioendothelioma
Metastatic disease most commonly originates from a primary colon, lung, breast or pancreatic cancer, and less commonly from less common tumors such as gastric , duodenal, carcinoid malignancies. Capsular metastases are most common form primary ovarian carcinoma.
Structural Principles Liver Cancer
Liver cancer arises from a single cell that for whatever reason assumes a different, and aggressive profile with an agenda and a time cycle that does not conform to the norm, but has the abillity to generate new cells of a similarly aggressive nature.
 Proliferation of a Malignant Cell with an Aberrant Sense of Time and An Aggressive Agenda Proliferation of a Malignant Cell with an Aberrant Sense of Time and An Aggressive Agenda |
| The image represents the life of a single set of cancer cells showing a progressioon of generations as the cell lives, dies and is regenerated. The orange products of the cell are seen in the background of the pink cytoplasm and the purple nucleus. The nucleus of the newest generation . Thecell is seen as a clock that has become distorted and time has become disordered. This process is abnormal and is a forerunner of a malignant process.
histology time malignancy cancer columnar cell histopathology Davidoff art copyright 2009 all rights reserved 85198j03s.81s |
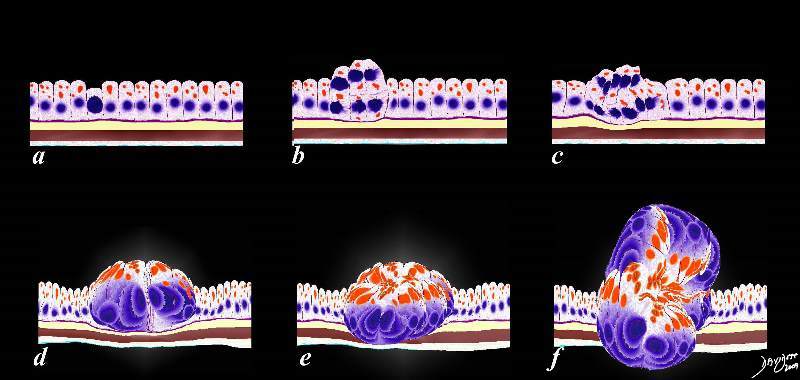 Abnormal Programming Multiplication and Space Occupation Abnormal Programming Multiplication and Space Occupation |
| The image represents the evolution of a single cancer cell (a) that fails to conform to the normal time cycle. In image a, the normal columnar mucosa is composed of rectangular cells supported by a thin purple basement membrane. One cell becomes aberrant, and is characterized by a large blue nucleus, scanty cytoplasm and a a change in it shape and overall appearance. The cell grows, multiplies, (b,c,d) and then invades the space of other parts of the tissue around it.
code histology time malignancy cancer columnar cell histopathology Davidoff art copyright 2009 all rights reserved 85198fc02.8s |
The structure of the liver cell and its organization as a tissue is quite unique so that although the end result of space occupation, and uncontrolled proliferation are common to other cancers, it is unique in many of its structural consequences as a result of unique architecture.
Structural Considerations
Normal Liver Histology
The parenchyma of the liver is a glandular epithelium, and the cells are aligned as plates or cords, radially arranged and separated by hepatic sinusoids making up a portal lobule and bounded by portal triads (radicles of the portal venous, hepatic arterial and bile duct systems).
The Cells
The dominant cell of the liver is the hepatocyte. The hepatocytes constitute approximately 70% of the liver by volume. It is therfore not surprising that most the primary liver cancers arise from this cell.
The hepatocyte is structurally characterised by its large size measuring between 20-30 microns, and the absence of a basement membrane, functionally characterised by their remarkable metabolic and regenerative capability. They characteristically respond to injury, whatever its type, by accumulating fat in the cytoplasm, an entity called steatosis.
|
The cell |
|
The cell is the building block of all biological structure. In this image a few almost hexagonal cells of the liver are attached together. Each cell has a central dark nucleus which is embedded in a pinkish cytoplasm. The nucleus takes up approximately 1/5 to 1/6 of the volume of the cell and usually lie in the center of the cell. Binucleate cells are normal and account for up to 25% of the cells. There is also considerable variation in the size of the nucleus from cell to cell. The red cells seen in the background are about 5 microns, and the liver cell is about 4 to 5 times their size. (Image courtesy of Barbara Banner M.D.) 13440 |
The Tissue
The liver is a compound tubular serous gland. The cells are aligned in cord formation that branch and anastomose alongside and in parallel with a vascular system called the sinusoids. These plates are arrranged like spokes of a wheel around a central vein which is a tributary of the hepatic veins. Kupffer cells are found within the space of Disse between the plates of cells and the sinusoids.
|
Cells Align and Combine in Cords of Tissue Separated by VAscular Channels Called Sinusoids |
|
The cells of the liver are organized in cords and plates and are distributed like spokes of a wheel around the central vein. The plates and cords(red overlay in b) are lined by the sinusoids (white channels between the cords) which are the channels which carry blood to and through the liver. Just below the sinusoids, between the wall of the sinusoid and the capsule of the liver there is a space called the space of Disse which carries the lymphatic fluid of the liver. Spindle shaped cells that are macrophages called Kupffer cells (ringed in blue) line the sinusoids and are related and integrated into the space of Disse.
liver hepatocyte cord plates sinusoids Kupffer cells macrophages defense blood flow histology normal (Image courtesy Barbara BAnner) copyright 2009 all rights reserved 13445c01.8s
|
The liver lobule is a structural and functional unit of the liver. At its centre is the central vein and it is bounded by several portal triads consisting of a hepatic arterial tributary, a portal venous branch, and a biliary radicle. The hepatic arteriole, and portal venule bring blood to the lobule and join to form the sinusoid that runs between the plates of hepatocytes. The biliary radicle takes bile away from the smaller branches of bile ductules that lie between the hepatocytes.The lobule is polyhedral in shapeand has a wheel like formation as described above with spokes of hepatocytes and sinusoids radially positioned around the central vein, and supported by connective tissue. It is functionally characterised by its ability to synthesis metabolically active substances and also by its ability to break down products (cataboloic activity), and by its ability to particpate in defense mechanisms and filtering mechanisms of the body.
The primary blood flow to the liver is via the portal vein which receives mesenteric blood flow which enters the lobules as the portal vein radicle in the portal triad. The radicles of the hepatic artery bring oxygenated blood to the liver, and bile ducts take bile to the duodenum via the gallbladder and common bile duct to facilitate fat digestion. Metastatic deposits that are hematogenously spread are therefore brought to the liver as a first stop if they are transported by the mesenteric veins. It is therefore no surprise that the liver is the most common site for metastatic colon carcinoma.
|
The Liver Lobule |
|
The sinusoids and hepatic cords combine to form a liver lobule which is a functional and structural unit of the liver. At the center of the lobule is the central vein (royal blue) from which emanate many cords of liver tissue. At the periphery of the lobule there are 4-5 groups of portal triads consisting of distal branches of the portal vein (dark blue), hepatic artery (red) and biliary radicle (green). They create the border of the lobule. (Image courtesy of Ashley Davidoff M.D.) 13009 W |
There are 3 zones within a liver lobule. Zone 1 is closest to the periphery of the lobule and portal triad, zone 2 is the intermediate zone, and zone 3 is in the centre of the lobule abutting the terminal hepatic venule. Zone 3 is the most vulnerable since it is furthest away from the oxygenated blood.
Each lobule measures 1-2mm in size, hexagonal in shape, is metabolically very active, and has tremendous regenerative capacity. The cells are exposed to gastrointestinal products of digestion that are absorbed into the blood, and traansported to the liver by the superior mesenteric vein which empties into the portal vein. Alcohol for example is transported to the liver via this system, and when taken in excess for long periods exposes the liver cells to its toxic effects causing cirrhosis, and eventually, in some patients resulting in dysplastic and finally to malignant neoplastic change.
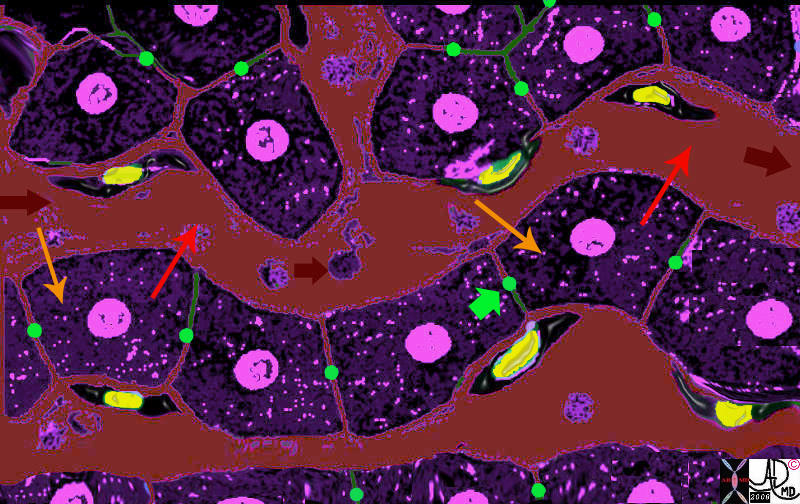
The Cells, The Plates, The Sinusoids and The Kuppffer Cells |
| The liver cells (purple) are aligned in plates, with bile canaliculi(green) running between them.· The Kupffer cells (yellow nuclii and black cytoplasm) run alonside the hepatocytes, and between the sinusoids (maroon) and the liver cells.· As the blood enters the sinusoids (maroon arrow) the cells absorb the products of digestion (yellow arrow) metabolise the products, and then export the new product back into the blood (orange arrow).· The Kuppffer cell acts as a macrophage and engulfs any foreign material, organisms,·or “debris” in the blood and acts as a defense mechanism.
13062b05b04.8 liver hepatocyte sinusoid Kupffer cell space of Disse bile canaliculus import export excrete transport portal blood hepatic arterial blood metabolism process receive export excrete Davidoff MD Davidoff art |
The sinusoids terminate at the central vein, each a part of the liver lobule. They converge with other central veins, to form the 3 major hepatic veins which transport the nutritionally rich blood to the right atrium via the inferior vena cava.

Lobules |
|
There are a multiplicity of lobules, each with a central vein and each delivering the “goods” to venules, which collectively join to form the hepatic veins and then into the IVC. Destination? The heart, from where it will be distributed to the body wide system. (Image courtesy of Ashley Davidoff M.D.) |
The Connective Tissue
The mesenteric extension of the lesser omentum to the liver is called the gastrohepatic ligament and it contains extensive lymphatic networks. The liver capsule also extends toward the porta hepatic to surround the portal triad. As they converge on the porta hepatics they create a sheath around the portal triad, and incorporate the lymphatic with them. As the vessels divide they carry with them a sheath of connective tissueenvelope.
Tumor that is within the lymphatics such as that from a primary gastric carcinoma will be swept within the lymphatics into th liver. Additionally it is also the route of spread of cancers that have spread into the peritoneum. Ovarian cancer is the primary and most common malignancy to spread this way.The capsule
The capsule that surrounds the portal triad structurally characterized being the intrahepatic extension of the liver capsule consisting of serosal outer and fibroelastic inner capsule. It also forms a loose capsule around the liver lobule. Functionally characterised as a support and protective barrier for the portal triad and the liver lobule serving to support the fine reticular network within the lobule disease from the peritoneal cavity may spread within the portal triad within the lymphatics, or within its spaces.
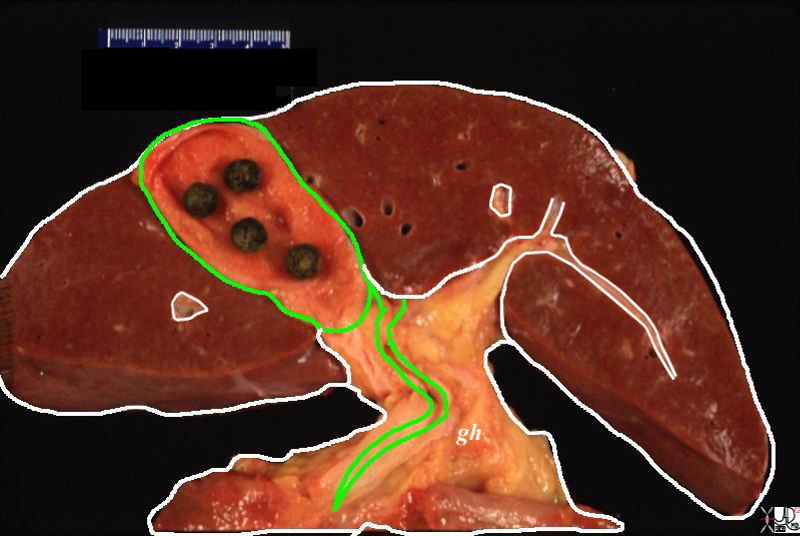
The Capsule of the Liver, Gastrohepatic Ligament, and Glissons Capsule |
| The gastrohepatic ligament (gh)connects the liver and the stomach not only as a ligamentous connection but also with lympfatics, as well as the portal triad ehich run on its free edge. In this diagram only the file duct is shown (green)It extends and connects to the liver capsule (white covering) around the liver, but also into and around the portal triads within the liver. These ligaments, with lymphatics are an important rourte of spread of disease such as gastric malignancy and ovarian malignancy which tends to spread transperitonealy.
44409b02.8s liver capsule gastrohepatic ligament Glisson’s capsule normal anatomy portal triad bile duct anatomy Courtesy Ashley DAvidoff MD copyright 2009 |
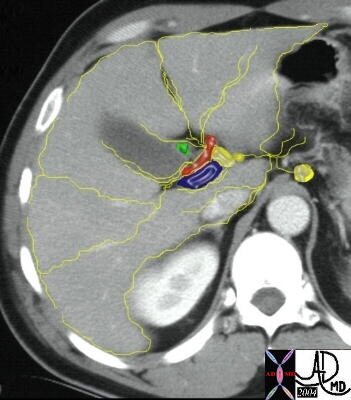
Lympahtic Connections Around the Portal Triad and Capsule |
| The lympatic drainage of the liver and connective tissue are intimately related, and is another map of how cancer of the ovary spreads into and around the liver from the peritoneal cavity.
24776 R W c liver + lymph nodes lymphatics+ lymphatic drainage portal triad + fx normal + anatomy + imaging radiology CTscan |
The connective tissue of the liver consists of fine reticular networks that bind the liver cells together in a delicate weave
·
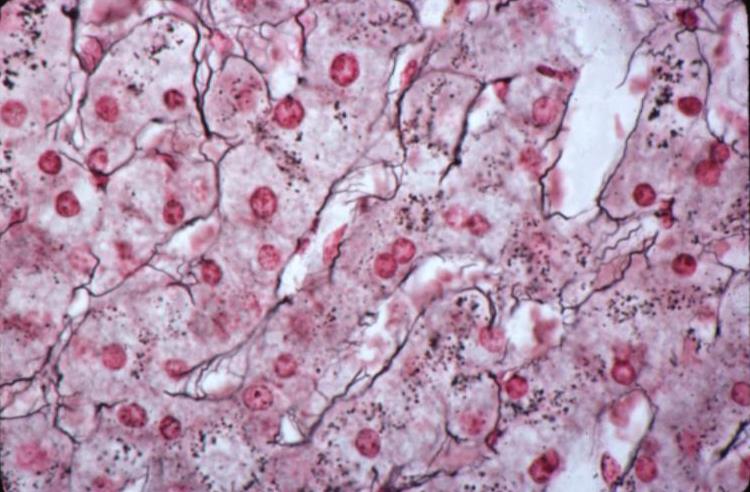
Reticular Fibers of the Liver |
| The fine reticular elements of the liver shown as black strands in this reticulin stain weave a fine but binding infrastructure of the liver cells.
13236 liver hepatocyte liver cords sinusoids cytoplasmic granules connective tissue reticulin histopathology histology Courtesy Barbara Banner MD |
Segments of the Liver
It is important to be familiar with the surgical anatomy of the liver since accurate positional descriptions of liver lesions are needed for the mapping of surgery, and determining operability and requires universal acceptance so that anatomists, pathologists, radiologists, hepatologists and surgeons are referencing the disease accurately.
There have been numerous methods of dividing and naming the parts of the liver. The earliest methods divided the liver into the left lobe, the quadrate lobe, the right lobe and the caudate lobe. Subsequently, the liver divisions were based on the venous anatomy. The right lobe was separated from the left by the middle hepatic vein. The falciform ligament divided the left lobe into the medial segment (closest to the middle hepatic vein) and the lateral segment. The right lobe was divided by the right hepatic vein into an anterior segment and a posterior segment. The next several diagrams outline the current segmental nomenclature which is still based on the distribution of the hepatic veins: Segment I = caudate lobe Segments II, III & IV = left lobe Segments V, VI, VII & VIII = right lobe
|
Segments of the liver |
|
This coronal image shows the division of the liver into right and left lobes, by the middle hepatic vein (blue), and the division of the right lobe into its superior and inferior segments and the division of the left by the cfalciform ligament (green) into its segments. (Image courtesy of Ashley Davidoff M.D.) |
|
Segments of the liver |
|
The following cross sectional image is viewed through the superior aspect of the liver with the hepatic venous system in color overlay. The right hepatic vein divides the right lobe into a posterior segment VII and an anterior segment VIII. The left hepatic vein divides the left lobe in the expected location of the falciform ligament into a rightward subsegment IVa and leftward II and III. (Image courtesy of Ashley Davidoff M.D.) |
The following image reveals the cross sectional segmental pattern at the level of the porta hepatis.
|

Segments of the liver |
|
Note that the falciform ligament anteriorly divides the left lobe. (Image courtesy of Ashley Davidoff M.D.) |
|
Segments of the liver |
|
This image demonstrates the royal blue IVC, posterior to the caudate lobe (I), the dark navy blue portal vein anteriorly, the green line and yellow dot indicating the position of the falciform ligament, the thick blue line of the middle hepatic vein and the thin blue line of the right hepatic vein. Since we are seeing the inferior aspect of the liver we are thus seeing segments V and VI of the right lobe, subsegment IVb and segments II and III of the left. (Image courtesy of Ashley Davidoff M.D.) |
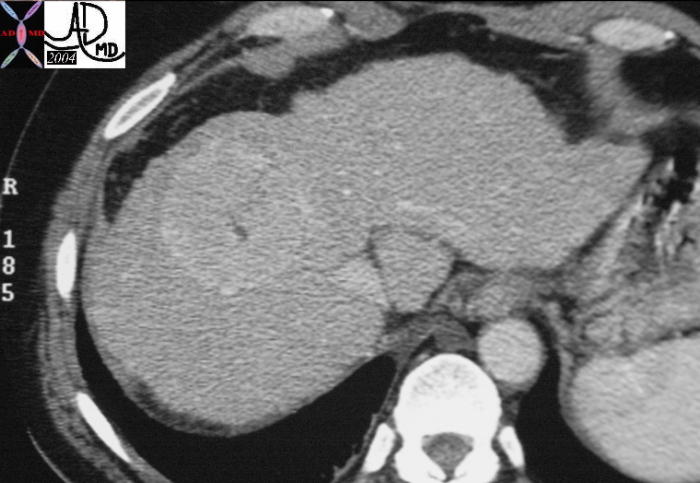
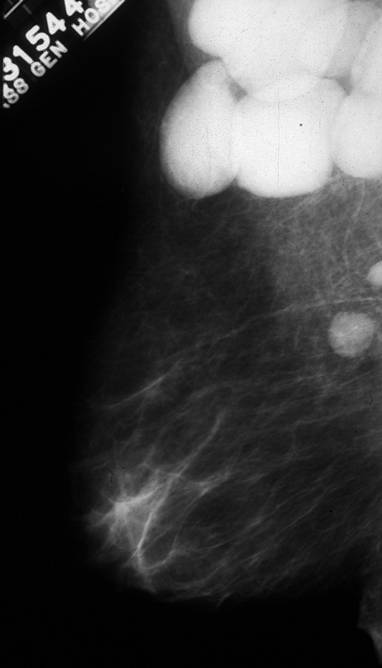
Segment VIII Liver Lesion |
|
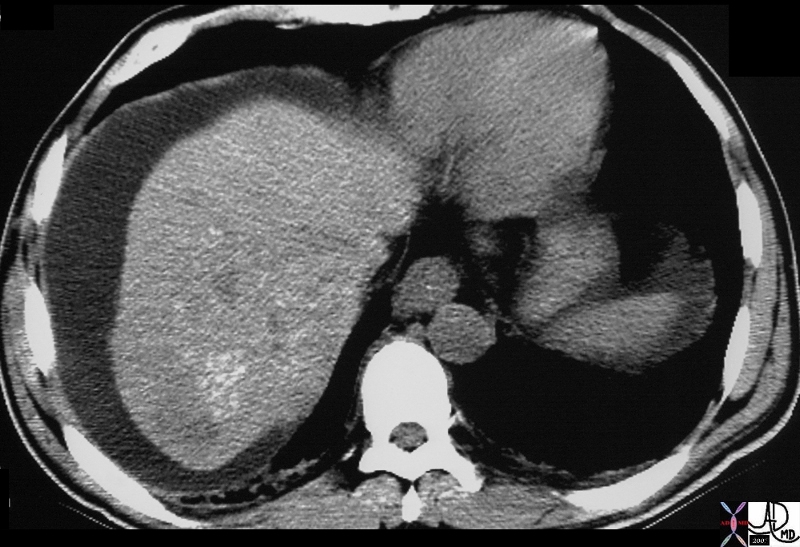
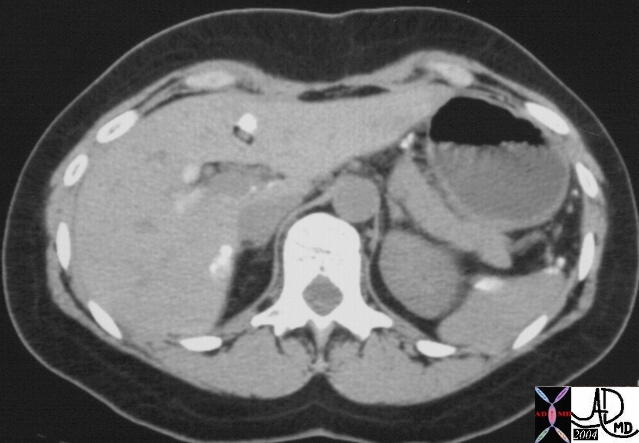
It is usual that in order to accurately locate the lesion that all the axial images have to be reviewed so that the relationship to all the veins is defined. 16487 liver hepatic fx mass fx hypervascular almost isodense HCC hepatoma hepatocellular carcinoma CTscan Courtesy Ashley Davidoff MD |
The caudate lobe (also known as segment I) is positioned medial to the right lobe and posterior to the left lobe. It is bounded inferiorly by the porta hepatis, posteriorly by the inferior vena cava, anteriorly by the portal vein and more superiorly and anteriorly by the by the ductus venosus. The caudate lobe is a rather curious structure, situated almost as an appendage to the liver. A unique characteristic is that its venous drainage flows directly and independently to the inferior vena cava below the diaphragm. As previously stated, it belongs neither to the left nor the right lobe, and it is considered as an “independent” segment.
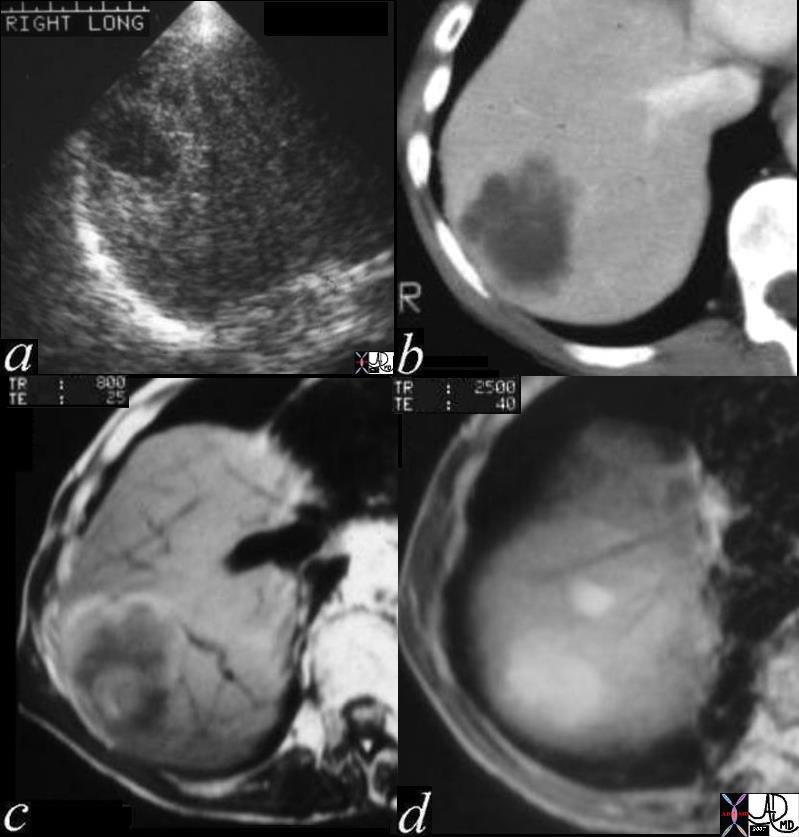
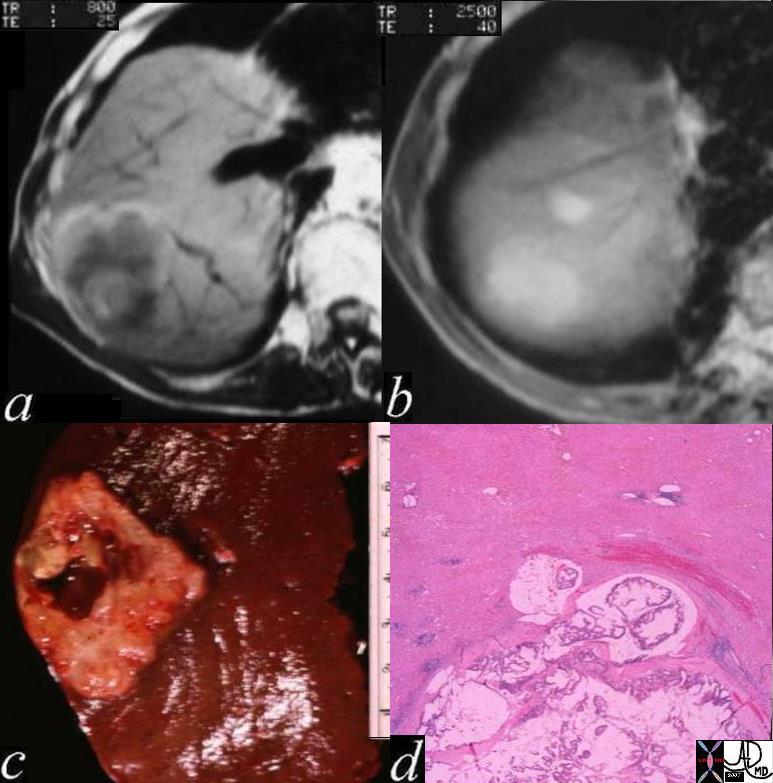
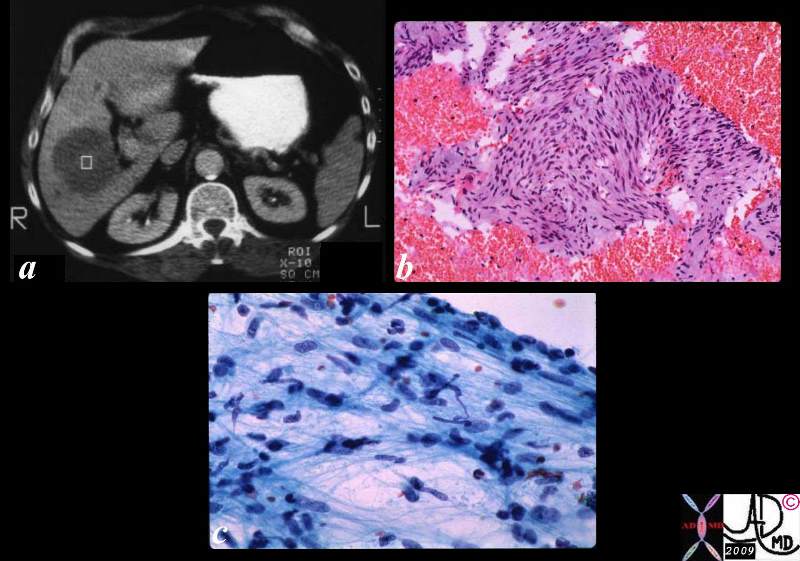
Hepatocellular Carcinoma
Hepatocellular (HCC)·carcinoma is a primary malignancy of hepatic parenchymal cells, characterized by space occupation and malignant proliferation of aberrant liver cells, with a tendency to invade veins,·with a·predisposition to multicentricity·that usually develops in patients with risk factors·of·alcohol abuse, viral hepatitis, and metabolic liver disease.
The disease is often clinically silent in the early stages unless it involves the capsule when it will resultin pain.·Continued clinical attention is paid to to·patients who have a history of alcoholoc cirrhossis, or a history of hepatitis b or c.· These patients are usually followed carefully, and are intermittantly screned for the development of hepatocellular carcinoma (hcc).· A rise in the serum level of alpha fetoprotein is particulalrly concerning.· As the tumor ot tumors advance in the liver, systemic symptoms such as malaise, fatigue, weight loss, fever of unknown origin, and hepatomegaly.··Less common presentations include hepatic rupture with hemoperitoneum.
The diagnosisis made by elevated serum alpha fetoprotein, or by imaging studies which include ultrasound, computed tomography (CT) and magnetic resonance imaging (MRI).··
Surgical resection and liver transplantation remain the mainstay of curative therapy. The results of the various medical treatments (chemotherapy, chemoembolization, ablation, and proton beam therapy) remain disappointing. Untreated HCC carries a poor prognosis and is directly related to tumour stage and degree of cirrhosis.
According to the classification by the International Working Party of the World Congress of Gastroenterology, hepatic nodules in patients with chronic liver diseases are subdivided into regenerative nodules (mono acinus and multi acinus), low-grade dysplastic nodules, high-grade dysplastic nodules, well-differentiated HCC, moderately- differentiated HCC, and poorly-differentiated HCC, in an ascending order of histologic grades, representing a sequence of multistep hepatocarcinogenesis.
Regenerative nodules represent focal proliferation of hepatocytes in response to various injurious stimuli. They can be classified as micronodular (<5 mm) or macronodular (>5 mm). Their blood supply is from the portal vein and hence they show an enhancement pattern similar to the normal liver parenchyma. Some nodules may contain iron (siderotic nodules) and these have a greater propensity towards dysplasia.
Dysplastic nodules are premalignant and contain atypical hepatocytes without definite features of malignancy on histology. According to the severity of the cellular atypia, they can be low grade or high grade and can undergo malignant transformation in a short duration of 4 months. They receive their blood supply from the portal vein and are hypovascular. However, occasionally they can have increased arterial flow.
HCC may be solitary, multifocal or diffusely infiltrative. Small HCC (<3 cm) are usually well differentiated whereas larger and diffuse HCC are poorly differentiated. They receive their blood supply mostly from the hepatic artery although rarely portal venous supply may be noted. Fibrous capsule and fat may be noted in well differentiated HCC thus differentiating them from dysplastic nodules. Fibrous septa, necrosis, hemorrhage and invasion of veins and bile ducts can also be present.
On gross pathology HCC may show 3 different patterns :
- Nodular form – single or multiple with or without a capsule
- Massive form -appears as an infiltrative mass in noncirrhotic livers
- Diffuse form – miliary infiltration of the liver parenchyma .
Cellular Changes
Histologically there are four different architectural types of HCC .
- Trabecular type – is the most common type
- Pseudo glandular type
- Solid
- Scirrhous or sclerosing type
Cytologically the cells of HCC exhibit varying degrees of hepatocellular differentiation. Histological criteria in favor of HCC include prominent nuclear atypia, high nuclear to cytoplasmic ratio with nuclear density twice normal, plates 3 or more cell thick , numerous unaccompanied arteries , mitoses in moderate numbers and invasion of stroma or portal tract .
Cause and Predisposing Factors
Cirrhosis from any cause is the seed-bed of HCC. The risk varies according to the aetiology of the cirrhosis.
- Hepatitis B Chronic infection with this virus is the most common cause of hepatoma worldwide.
- Hepatitis C As potent an oncogenic virus as HBV but less prevalent. Usually requires 25 years of chronic infection to cause tumour.
- Alcoholic liver disease Oncogenic potential probably mediated by inflammation and cirrhosis. May be augmented by concurrent viral infection.
- Haemochromatosis -Iron overload, Untreated genetic haemochromatosis is an especially severe premalignant state.
- Tyrosinaemia HCC occurs in 37% of patients who survive to 2 years of age and may occur in patients who have had successful liver transplants
- Oral contraceptives Absolute risk of developing HCC very small.
- Anabolic steroids Case-reports suggest a small increased risk for HCC
Genetic
Chromosomal abberations have been frequently reported in HCC.· Amplifications of the chromosomes are noted in 1q, 8q, 6p, and 17q. Among the chromosomes most frequently lost in HCC are 8p, 16q, 4q, 17p, and 13q. These chromosomal regions contain key players in hepatocarcinogenesis such as p53 (chromosome 17p) or Rb (chromosome 13q).
Genes involved in important regulatory pathways are the targets of these mutations. Four main pathways can be distinguished
• The p53 pathway involved in DNA damage response and apoptosis,
•The b-catenin/APC pathway involved in intercellular interactions and signal transduction
•The TGF-b pathway involved in growth inhibition and hepatocyte death
• The RB1 pathway involved in cell cycle control.
Connections between these different pathways are most probable.
·
Carcinogens
Plant carcinogens: Aflatoxins produced by fungi Aspergillus flavis and A. parasitans are well recognized to cause severe hepatic injury.These fungi grow readily on grains, peanuts, and food products in the humid subtropical and tropical regions.Higher HCC mortality rates have also been found in people who drink pond-ditch water contaminated with the blue-green algal toxin microcystin, the toxin also causes hepatic hemorrhage and necrosis.
Chemical carcinogens :DDT,Nitrites, hydrocarbons, solvents, organochlorine pesticides, primary metals, and polychlorinated biphenyls have been implicated as potential carcinogens.
Pathophysiology and Pathogenesis
Result
Potential Complications
- Natural History
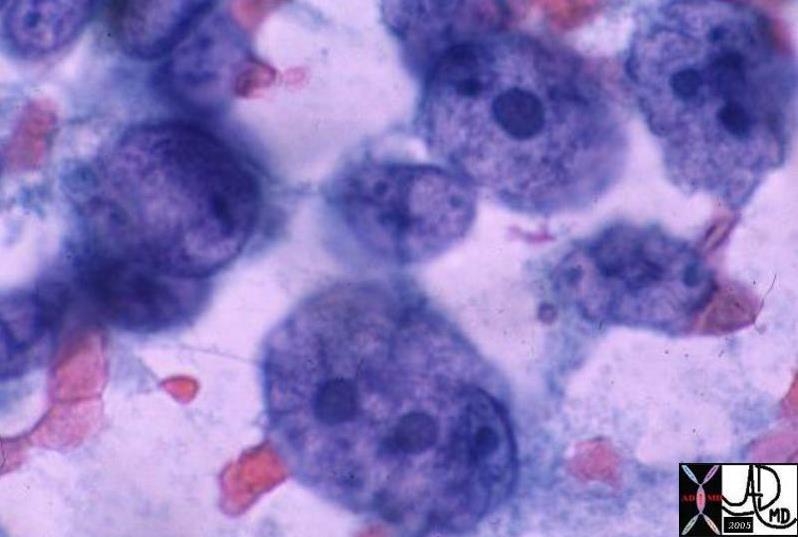 03539 03539 |
| 03539 liver hepatocytes fx enlarged HCC hepatocellular carcinoma hepatoma histopathology Courtesy Ashley Davidoff MD |
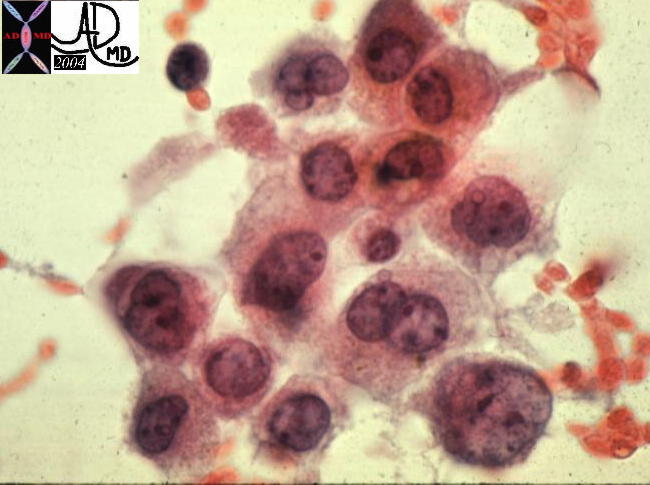 13447 13447 |
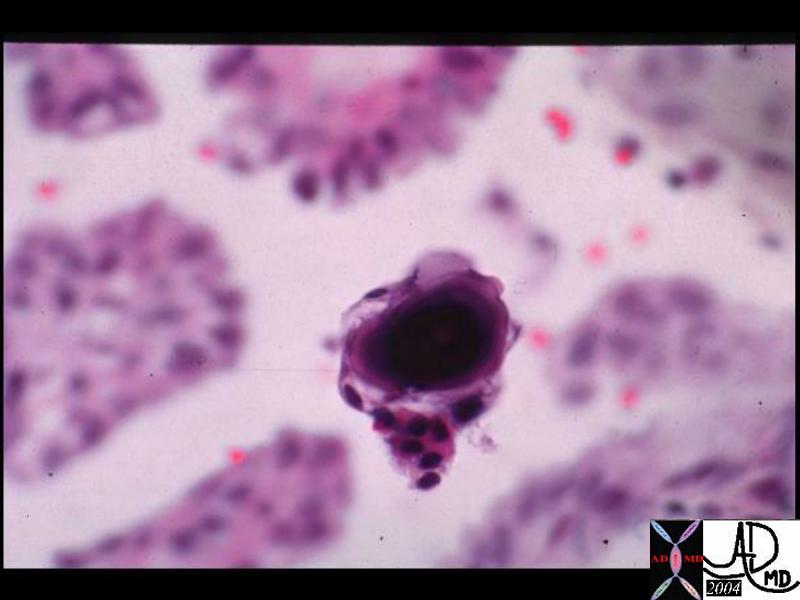
| 13447 liver HCC hepatocellular carcinoma cytopathology |
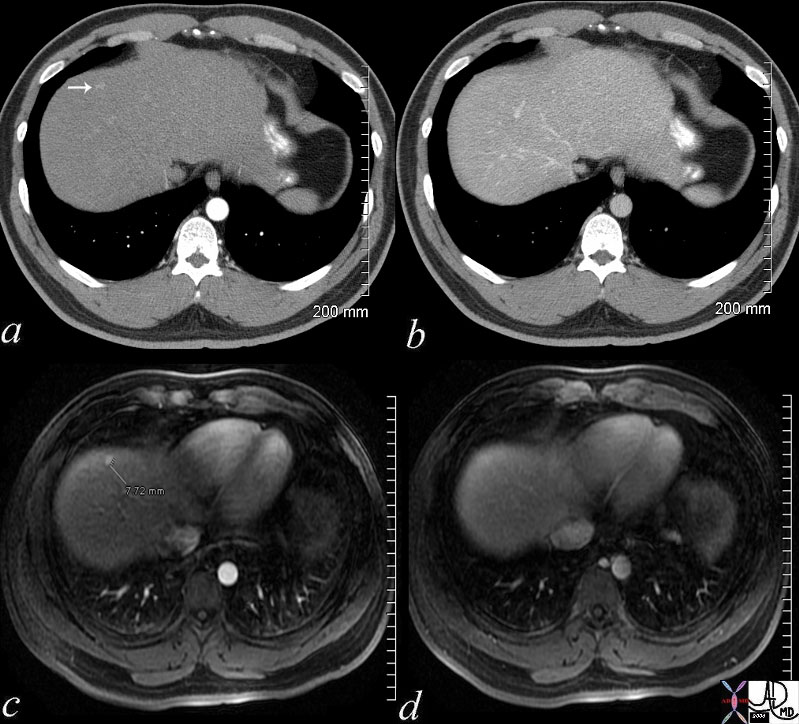 48369c03 48369c03 |
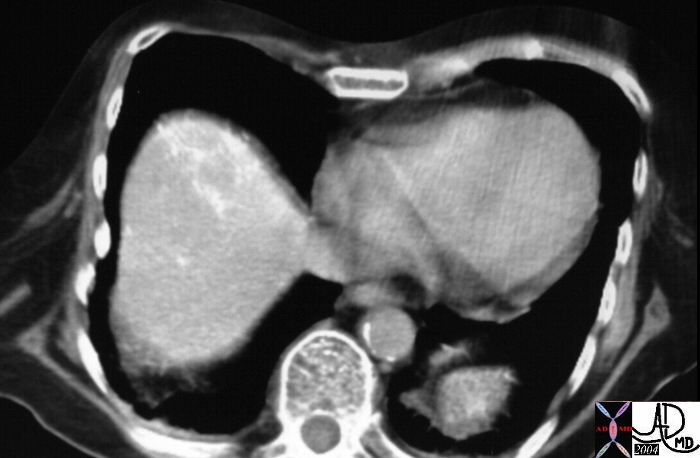
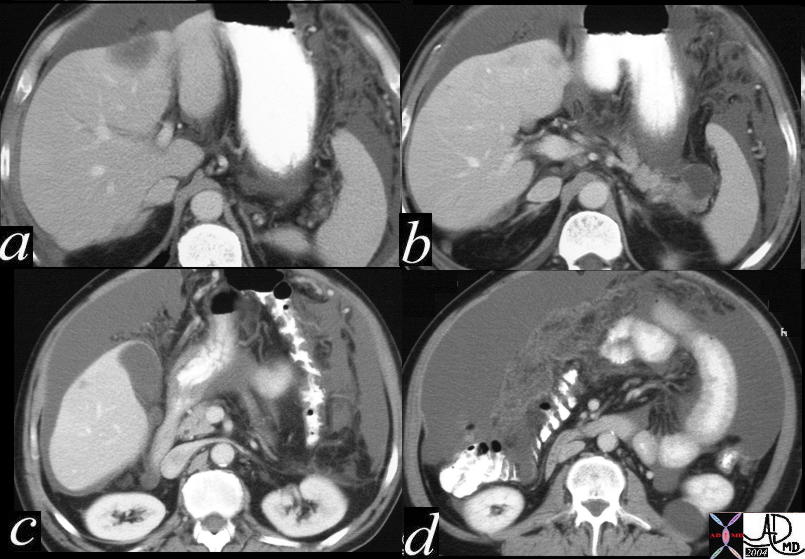
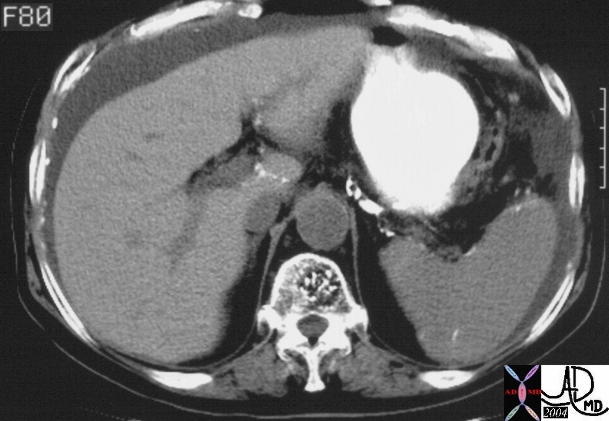
| 48369c03 40 male with hepatitis B liver fx hypervascular lesion seen in early arterial phase only with rapid wasout dx HCC hepatoma hepatocellular carcinoma capillary hemangioma characterisation characterization blood flow CTscan MRI Courtesy Ashley DAvidoff MD |
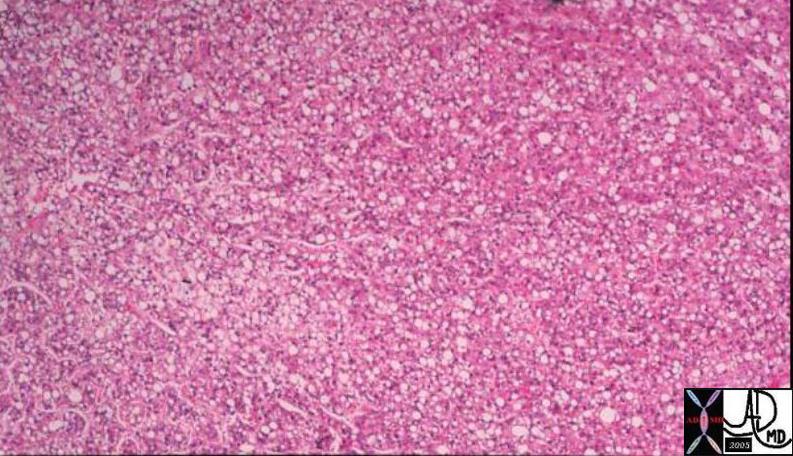
03503 |
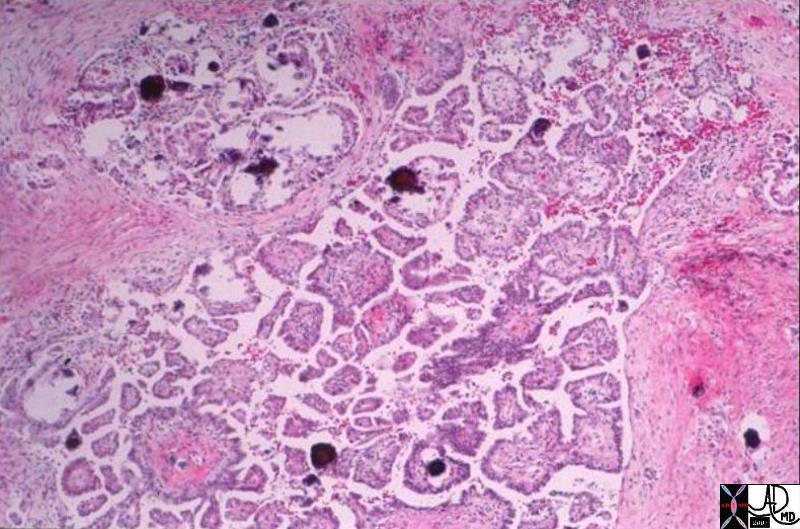
| 03503 liver mass fx fat fatty change within the lesion intracytoplasmic fat HCC hepatocellular carcinoma hepatoma histopathology Courtesy Barbara Banner MD |
·
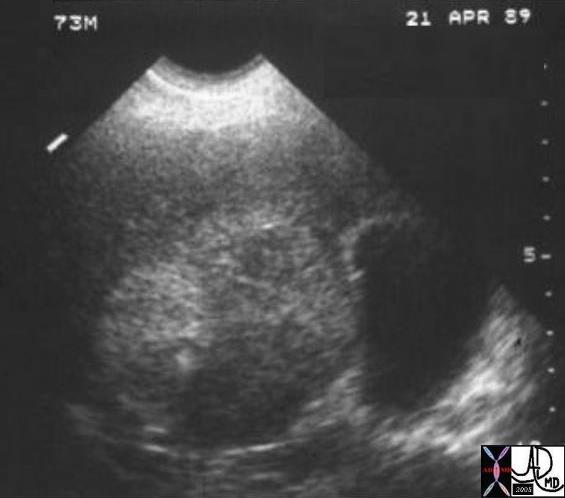
03760 |
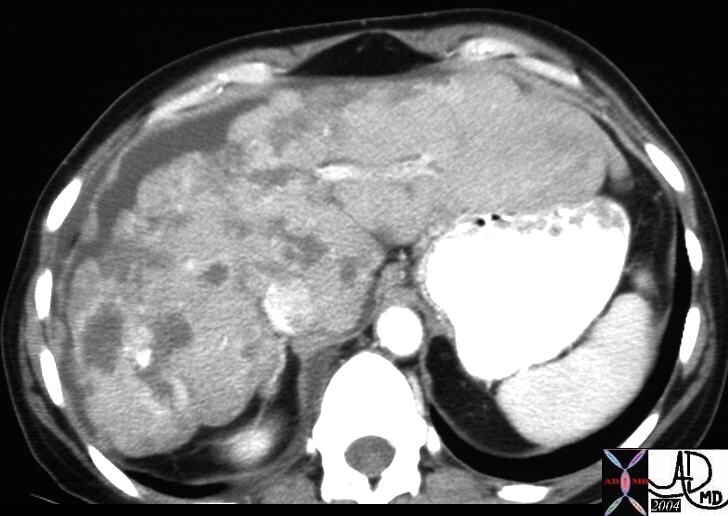
| 03760 liver fx hepatic mass echogenic heterogeneous HCC hepatocellular carcinoma hepatoma dx hemochromatosis dx fat in the lesion USscan Courtesy Ashley Davidoff MD |
·

03762 |
| 03762 liver fx hepatic mass fx question capsule lobular contours HCC hepatocellular carcinoma hepatoma dx hemochromatosis iron overload hemosiderosis grosspathology Courtesy Barbara Banner MD |
·
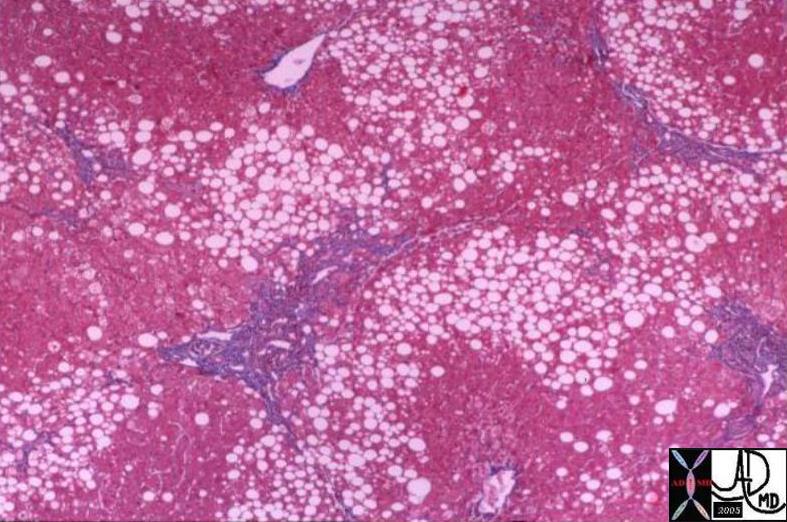
03763 |
| 03763 liver fx hepatic mass fx fat steatosis HCC hepatocellular carcinoma hepatoma dx hemochromatosis iron overload hemosiderosis histopathology Courtesy Barbara Banner MD |
·
·
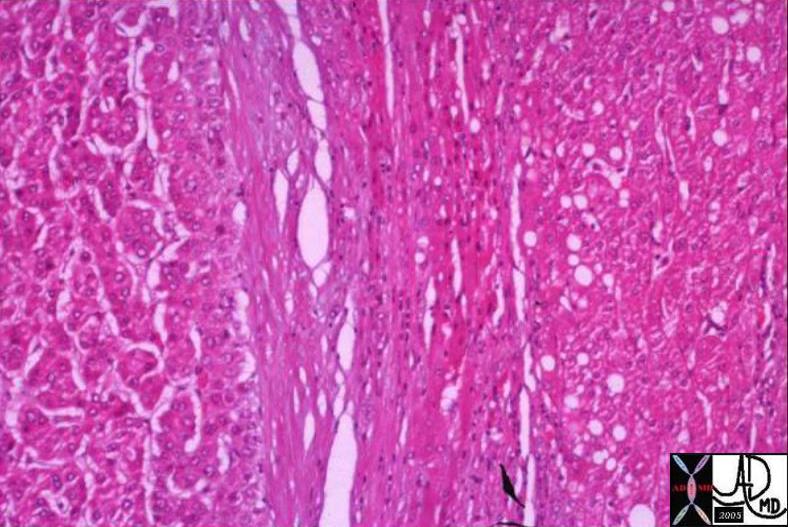
03764 |
| 03764 liver fx hepatic mass fx fat fx capsule steatosis HCC hepatocellular carcinoma hepatoma dx hemochromatosis iron overload hemosiderosis histopathology Courtesy Barbara Banner MD |
·
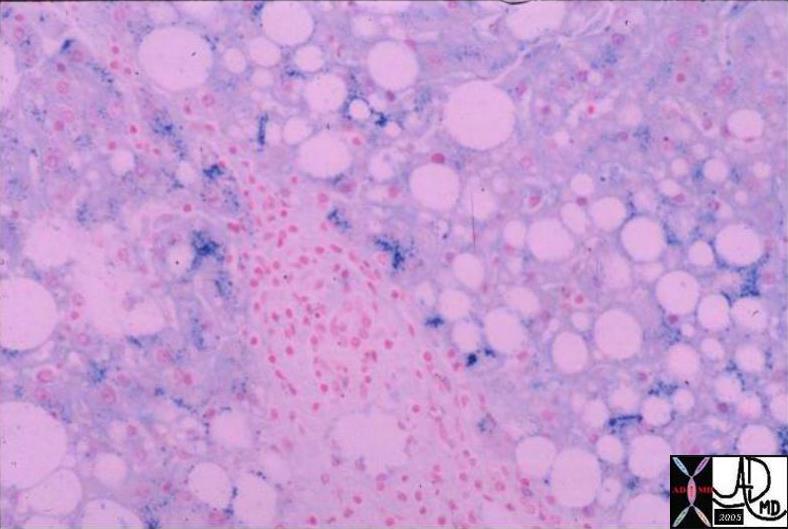
03765 |
| 03765 liver fx hepatic mass fx fat fx positive for iron steatosis HCC hepatocellular carcinoma hepatoma dx hemochromatosis iron overload hemosiderosis histopathology Courtesy Barbara Banner MD |
·
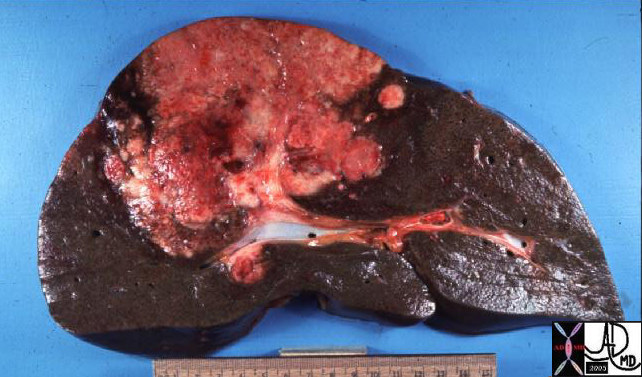
24008 |
| 24008 liver fx mass portal vein fx invasion satellite nodule multicentric dx hepatocellular carcinoma hepatoma HCC grosspathology Courtesy Barbara Banner MD |
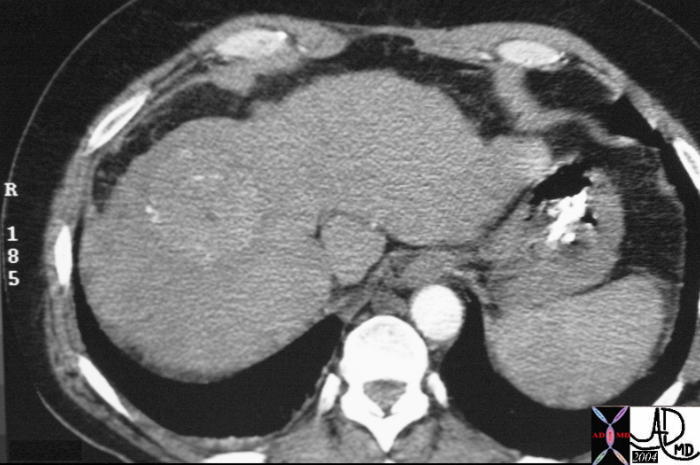
16486 |
| 16486 liver + fx mass + hypervascular arterial phase HCC hepatocellular carcinoma + imaging radiology CTscan C+ |
·
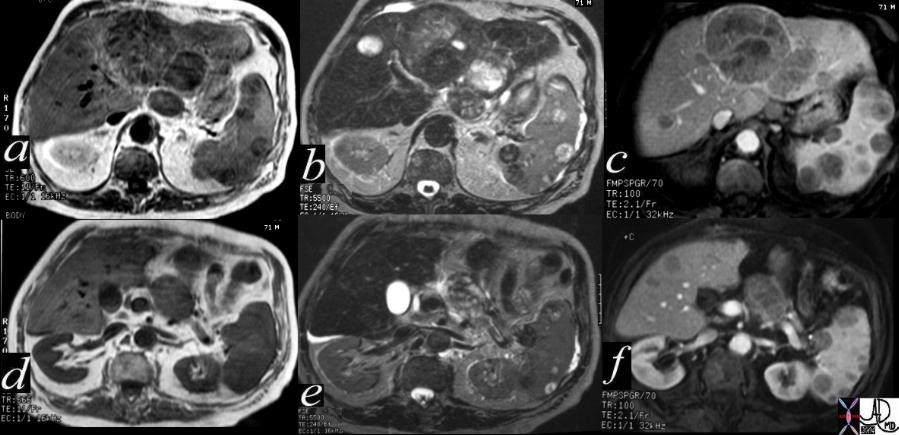
40417c |
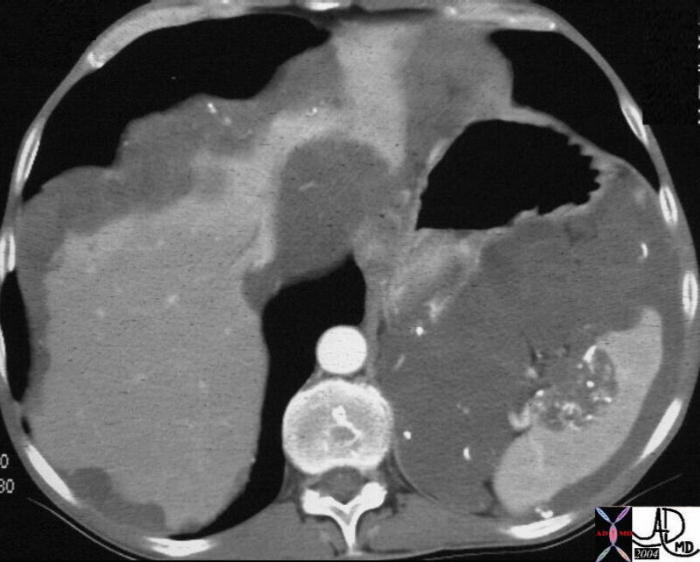
| 71 year old male with chronic alcoholism 40417c Courtesy Ashley Davidoff MD code liver left lobe fx mass code spleen fx multiple masses code pancreas pancreatic body fx mass code dx primary HCC hepatoma hepatocellular carcinoma complicated by metatstatic disease to the spleen and pancreas imaging radiology MRI T1 T2 neoplasm malignant carcinoma tumor cancer |
·
·
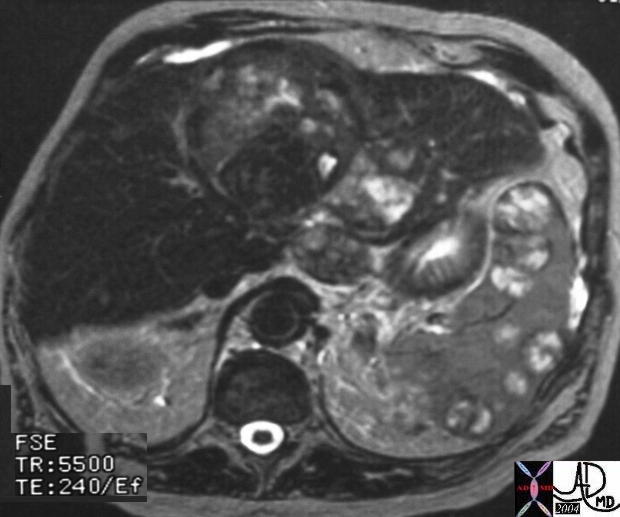
20860 |
| 20860 spleen + fx heterogeneous masses nodules water T2 bright nodules masses enhancement dark dx HCC metastasis primary liver irregula cirrhosis mass + hepatocellular carcinoma + imaging radiology MRI T2 neoplasm malignant cancer tumor |
·
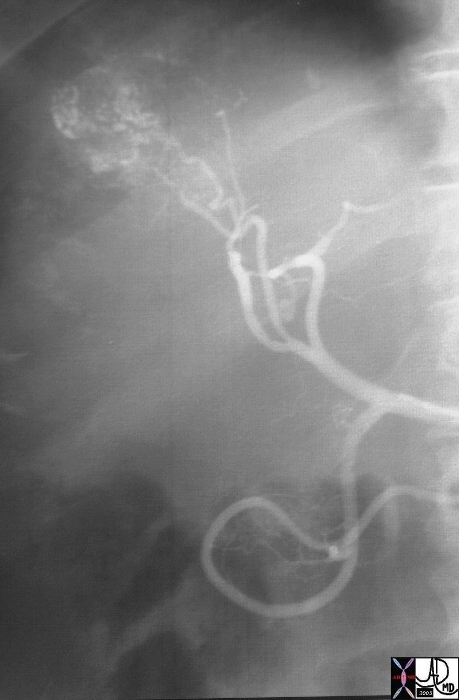
19675 |
| 19675 liver mass fx hypervascular mass early arterial phase arteriovenous shunting A-V shunting hepatpoma HCC hepatocellular carcinoma angiogram arteriogaphy angiography Courtesy Ashley Davidoff MD |
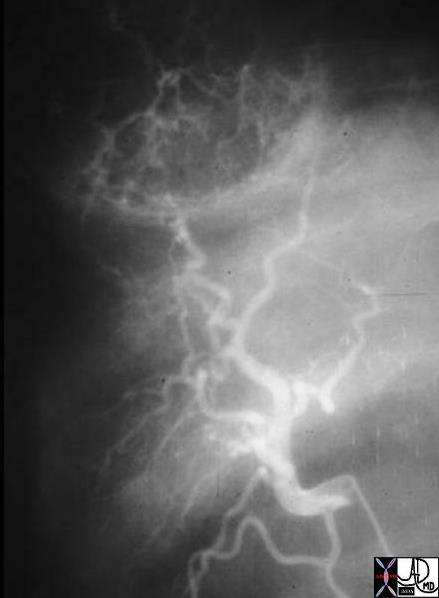
03552 |
| 03552 liver fx hypervascular mass HCC hepatocellular carcinoma hepatoma angiogram amgiography Courtesy Ashley Davidoff MD |
·
·
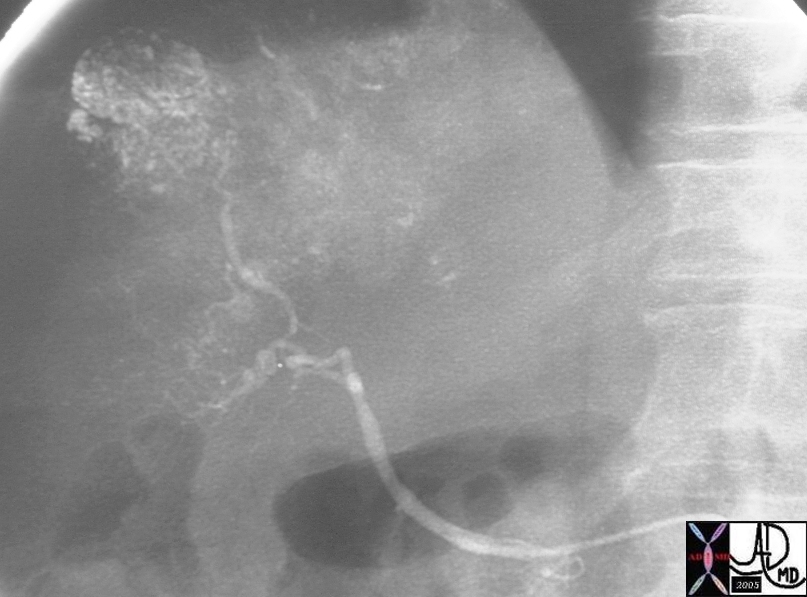
19681 |
| 19681 liver mass fx hypervascular mass early arterial phase hepatpoma HCC hepatocellular carcinoma angiogram arteriogaphy angiography ethiodol injection fx ethiodol accumulation Courtesy Ashley Davidoff MD |
·
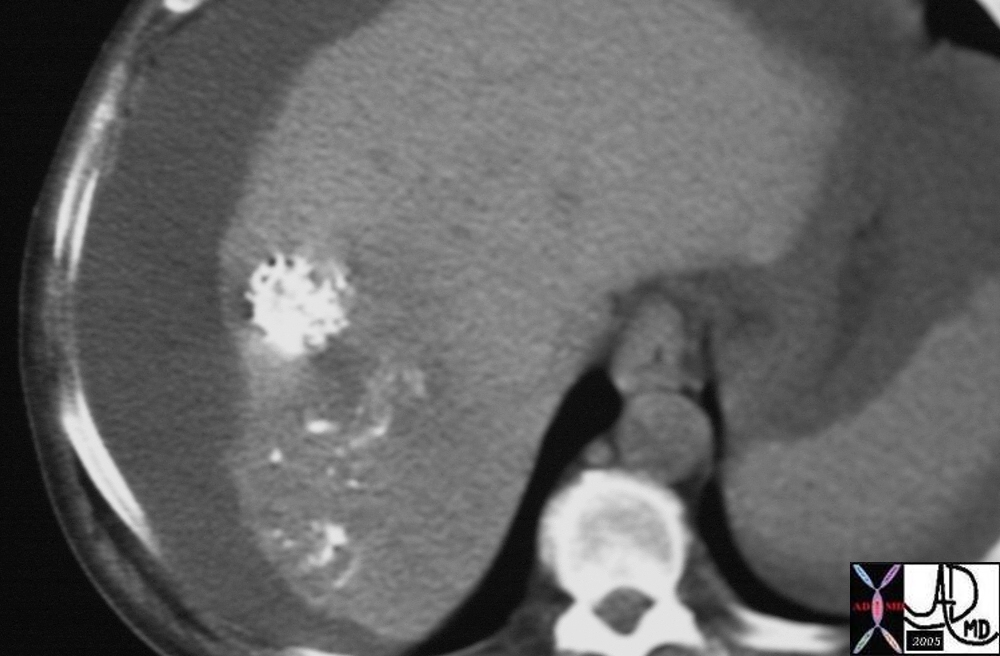
19678 |
| 19678 liver fx mass post ethiodol injection fx ethiodol accumulation hepatpoma HCC hepatocellular carcinoma CTscan Courtesy Ashley Davidoff MD |
·
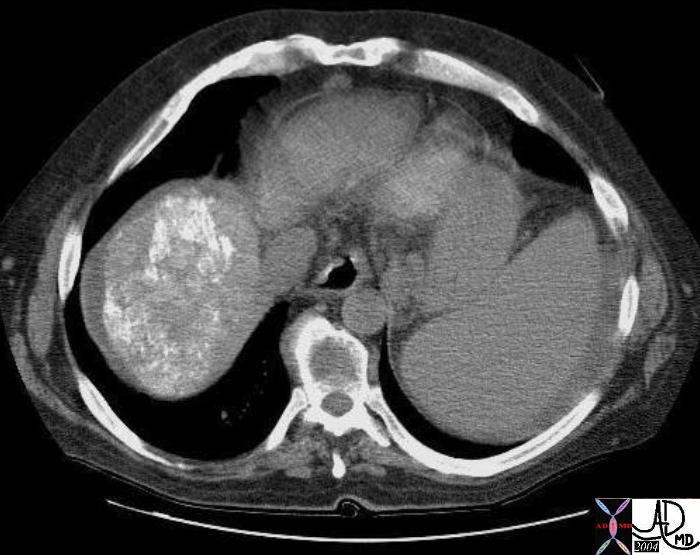
19543 |
| 19543 liver + fx mass + malgnant ethiodol + imaging radiology CTscan neoplasm malignant cancer tumor |
·
·
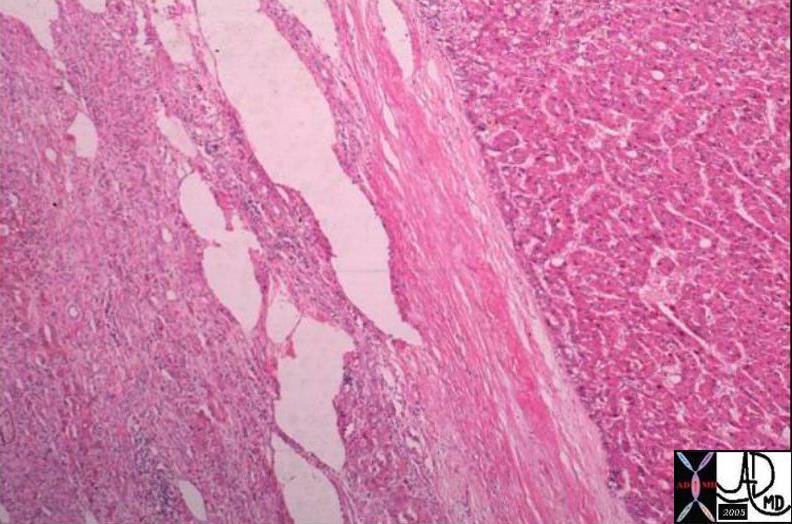
03751 |
| 03751 liver fx mass fx capsule HCC hepatocellular carcinoma hepatoma histopathology Courtesy Barbara Banner MD |
·
·
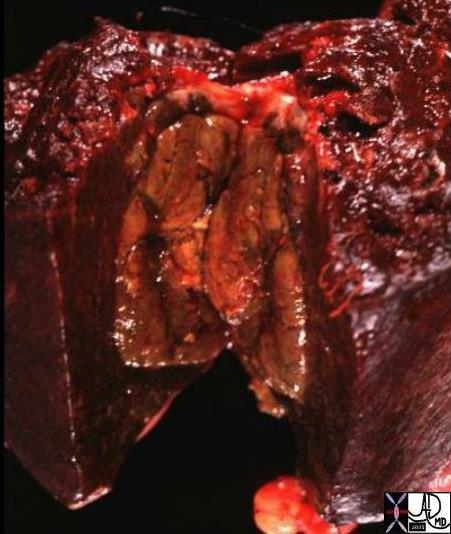
03750 |
| 03750 liver fx hepatic mass HCC hepatocellular carcinoma hepatoma grosspathology Courtesy Ashley Davidoff MD |
·
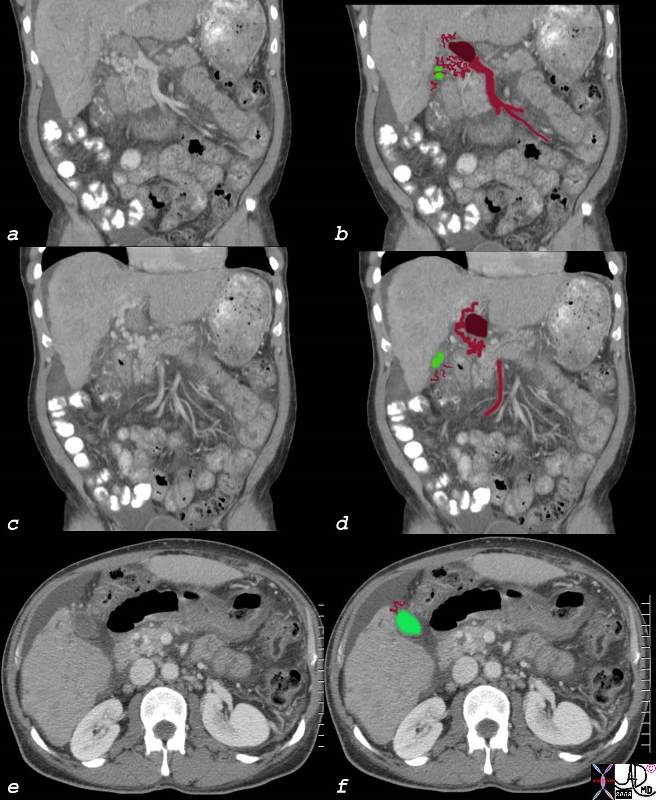
82709c01.8s |
| 82709c01.8s 60M liver HCC hepatocellular carcinoma portal vein thrombosis hepatic vein thrombosis cavernous transformation of the portal vein gallbladder varicosity ascites CTscan Courtesy Ashley Davidoff MD copyright 2008 CTscan |
·

03239 |
| 03239 liver hepatocellular carcinoma dx HCC infiltating infiltrative grosspathology Courtesy Ashley Davidoff MD |
·
03240 |
| 03240 liver hepatocellular carcinoma hepatocellular carcinoma dx HCC infiltating infiltrative dx HCC |
·
03486 |
| 03486 liver HCC infiltrative infiltrating hepatocellular carcinoma hepatoma Courtesy Ashley Davidoff MD |
·
03492 |
| 03492 liver HCC hepatocellular carcinoma infiltrative infiltrating hepatoma nuclear medicine NM liver spleen technetium scan |
·
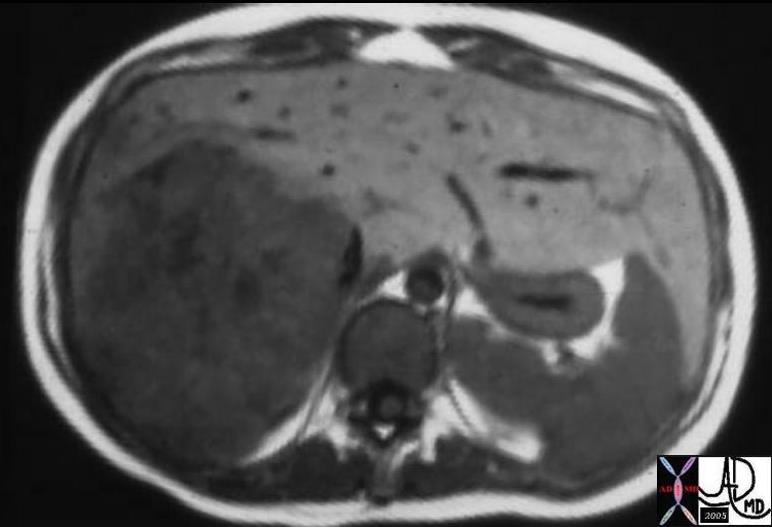
03573 |
| 03573 liver fx hepatic mass fx capsule HCC hepatocellular carcinoma hepatoma MRI T1 Courtesy Ashley Davidoff MD |
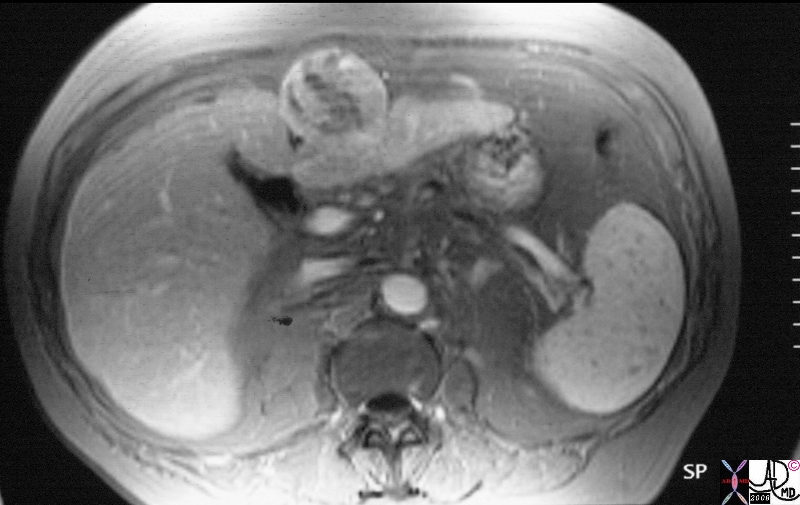
22855.8s |
| 22855.8s liver mass hepatoma hepatocellular carcinoma HCC gallstones cirrhosis Gamma Gandy bodies MRI T1 weighted with gadolinium fat suppression Copyright 2008 Courtesy Ashley Davidoff |
·
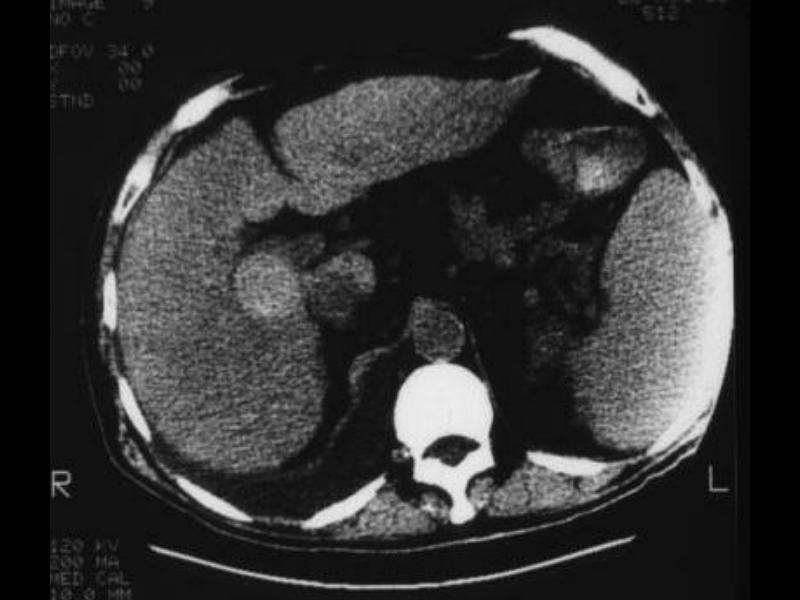
03274 |
| 03274 liver fx mass fx dense dx hemorrhage hemorrhagic hepatocellular carcinoma dx HCC CTscan Courtesy Ashley Davidoff MD |
·
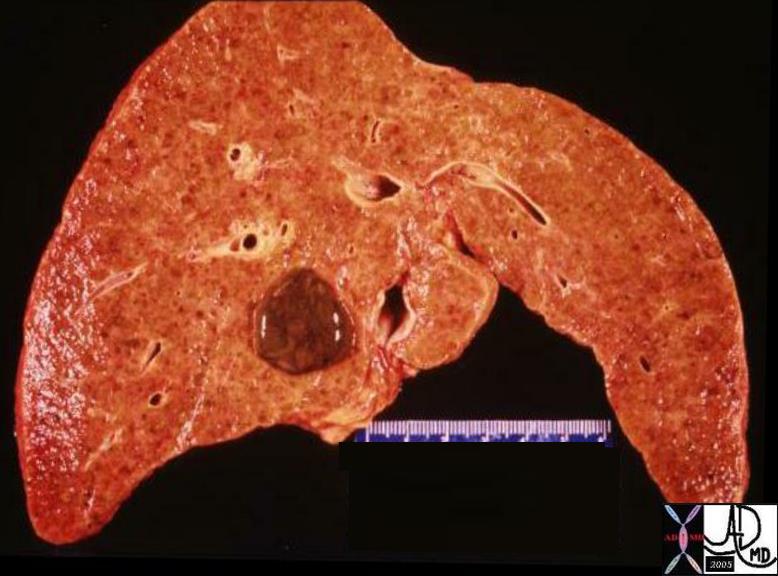
03271 |
| 03271 liver hepatocellular carcinoma dx HCC hemorrhagic nodule grosspathology Courtesy Ashley Davidoff MD |
·
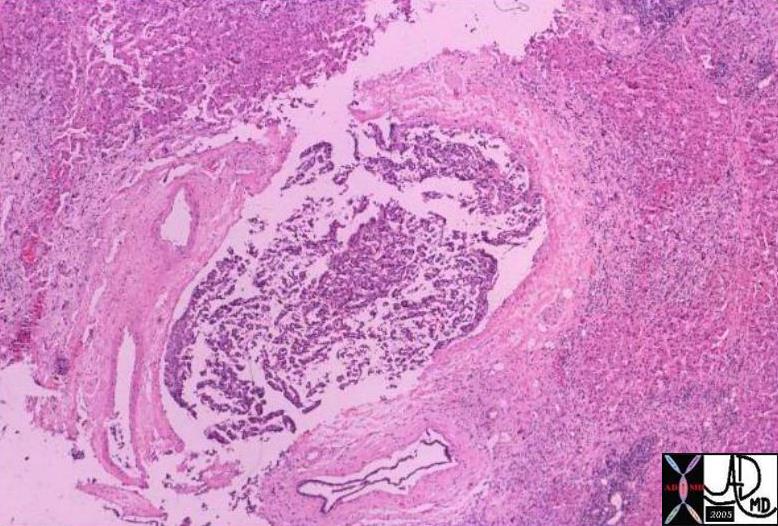
03463 |
| 03463 liver fx mass portal vein fx invasion dx HCC hepatocellular carcinoma hepatoma histopathology Courtesy Barabara Banner MD |
·
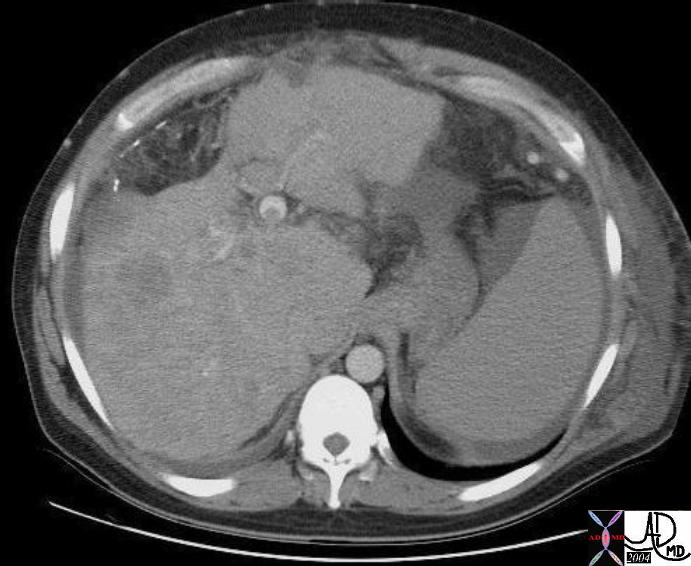
19547 |
| 19547 liver + vein + portal invasion portal vein thrombosis multicentric hcc + hepatocellular carcinoma liver fx irregular dx cirrhosis spleen mild enlargement splenomegaly abdomen abdominal ascites imaging radiology CTscan neoplasm primary carcinoma malignant cancer tumor |
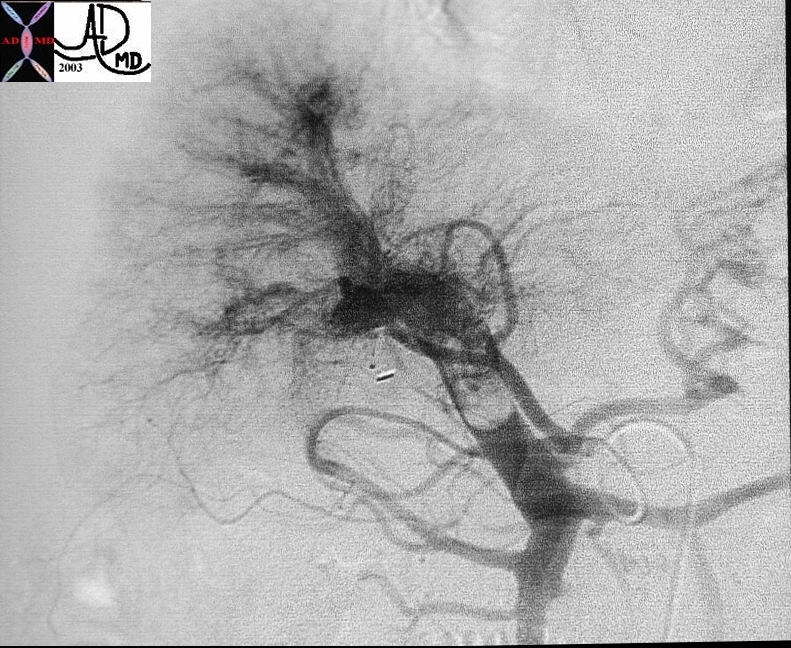
22446 |
| 22446 liver hepatic artery portal vein fx arterial portal shunting fx retrograde filling of the portal vein fx portal vein thrombosis PVT dx hepatocellular carcinoma HCC hepatoma angiography Courtesy Ashley Davidoff MD |
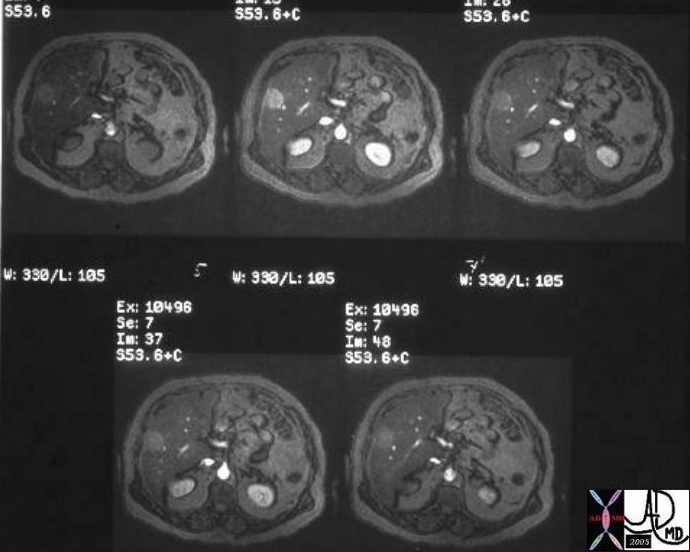
03427 |
| 03427 60 M liver hemachromatosis iron overload fx mass enhancing dx HCC hepatocellular carcinma clear cell Courtesy Ashley Davidoff MD |
Natural History
HCC presents in three principal ways
- A right-upper- quadrant mass,
- A worsening of the general health of a patient with cirrhosis
- Asymptomatically as a result of a radiological examination
·
·
·
Diagnosis·
·
·
Clinical Approach
·
Presentation depends on the stage of disease. In countries with systematic screening programs (Taiwan, Hong Kong, Japan and Korea) HCC is often diagnosed at an early stage when patients are asymptomatic or have symptoms due only to the underlying disease. In the United states, there is no systematic screening for HCC, patients usually present at a late stage, often with abdominal pain, palpable RUQ mass, weight loss, weakness, abdominal fullness and swelling – ascites, and nausea. Suspicion for HCC should be heightened when patients with compensated cirrhosis develop signs of decompensation. Jaundice due to obstruction of the intrahepatic ducts by the underlying liver disease or rarely due to hemobilia is relatively uncommon but ominous. Abdominal swelling may occur as a consequence of ascites due to the underlying chronic liver disease or may be due to a rapidly expanding tumor. In fewer than 5% of cases, central necrosis or acute hemorrhage into the peritoneal cavity leads to death. Hematemesis may occur due to esophageal varices from the underlying portal hypertension. Bone pain is seen in 3-12% of patients, but necropsies show bone metastases in ~20% of patients.
The triad of abdominal pain, weight loss and an abdominal mass is the most common clinical presentation in the United States.
·
Physical Signs Hepatomegaly is the most common physical sign, occurring in 50-90% of patients. Abdominal bruits are noted in 6-25%, and ascites occurs in 30-60% of patients. Ascites should be examined by cytology. Splenomegaly is mainly due to portal hypertension. Weight loss and muscle wasting are common, particularly with rapidly growing or large tumors. Fever is found in 10-50% of patients, from unclear cause. The signs of chronic liver disease may be present, including jaundice, dilated abdominal veins, palmar erythema, gynecomastia, testicular atrophy, and peripheral edema. Budd-Chiari syndrome can occur due to HCC invasion of the hepatic veins; it should be suspected in patients with tense ascites and a large tender liver.
·
Paraneoplastic Syndromes Most paraneoplastic syndromes in HCC are biochemical abnormalities without associated clinical consequences. They include hypoglycemia (also caused by end-stage liver failure), erythrocytosis, hypercalcemia, hypercholesterolemia, dysfibrinogenemia, carcinoid syndrome, increased thyroxin-binding globulin, changes in secondary sex characteristics (gynecomastia, testicular atrophy, and precocious puberty), and porphyria cutanea tarda. Mild hypoglycemia occurs in rapidly growing HCC as part of terminal illness, and profound hypoglycemia may occur, although the cause is unclear. Erythrocytosis occurs in 3-12% of patients, and hypercholesterolemia in 10-40%. A high percentage of patients have thrombocytopenia or leukopenia not caused by cancer infiltration of bone marrow, as in other tumor types.
·
Patients with HCV infection are more often screened and thus tend to present with signs and symptoms of cirrhosis and earlier stage HCC tumors. Patients with HBV infection or no serological evidence of hepatitis infection tend to present with larger tumors and less cirrhosis.
History
The history is important in evaluating putative predisposing factors, including a history of hepatitis or jaundice, blood transfusion, or use of intravenous drugs. A family history of HCC or hepatitis should be sought, and a detailed social history taken to include job descriptions for industrial exposure to possible carcinogenic drugs as well as contraceptive hormones.
·
·
Surveillance for Hepatocellular Carcinoma (HCC)
- Ø Patients at high risk for developing HCC should be entered into surveillance programs (Level I). The at-risk groups are identified in the table below.
- Ø Patients on the transplant waiting list should be screened for HCC because in the USA the development of HCC gives increased priority for orthotopic liver transplantation (OLT), and because failure to screen for HCC means that patients may develop HCC and progress beyond listing criteria without the physician being aware (Level III).
- Ø Surveillance for HCC should be performed using ultrasonography (Level II).
- Ø Alphafetoprotein (AFP) alone should not be used for screening unless ultrasound is not available (Level II).
- Ø Patients should be screened at 6 to 12 month intervals (Level II).
- Ø The surveillance interval does not need to be shortened for patients at higher risk of HCC (Level III).
·
·
·
·Surveillance Is Recommended for the Following Groups of Patients (Level III)
| Hepatitis B carriers |
For non-cirrhotic hepatitis B carriers not listed above the risk of HCC varies depending on the severity of the underlying liver disease, and current and past hepatic inflammatory activity. Patients with high hepatitis B virus (HBV) deoxyribonucleic acid (DNA) concentrations and those with ongoing hepatic inflammatory activity remain at risk for HCC. |
| Non-hepatitis B cirrhosis |
|
- Ø Nodules found on ultrasound surveillance that are smaller than 1 cm should be followed with ultrasound at intervals from 3 to 6 months (Level III). If there has been no growth over a period of up to 2 years, one can revert to routine surveillance (Level III).
- Ø Nodules between 1-2 cm found on ultrasound screening of a cirrhotic liver should be investigated further with two dynamic studies, either computed tomography (CT) scan, contrast ultrasound or magnetic resonance imaging (MRI) with contrast. If the appearances are typical of HCC (i.e., hypervascular with washout in the portal/venous phase) in two techniques the lesion should be treated as HCC. If the findings are not characteristic or the vascular profile is not coincidental among techniques the lesion should be biopsied (Level II).
- Ø If the nodule is larger than 2 cm at initial diagnosis and has the typical features of HCC on a dynamic imaging technique, biopsy is not necessary for the diagnosis of HCC. Alternatively, if the AFP is >200 ng/mL biopsy is also not required. However, if the vascular profile on imaging is not characteristic or if the nodule is detected in a non-cirrhotic liver, biopsy should be performed (Level II).
- Ø Biopsies of small lesions should be evaluated by expert pathologists. If the biopsy is negative for HCC patients should be followed by ultrasound or CT scanning at 3 to 6 monthly intervals until the nodule either disappears, enlarges, or displays diagnostic characteristics of HCC. If the lesion enlarges but remains atypical for HCC a repeat biopsy is recommended (Level III).
Diagnostic criteria
- Ø The presence of a nodule larger than 2 cm with hypervascular characteristics detected by at least two imaging techniques confirms the presence of HCC, with no need for cyto-histopathological confirmation.
- Ø AFP > 200 ng/ml plus an arterial enhancing lesion by one of the imaging tests meet criteria for the diagnosis of HCC.
Imaging
Imaging has numerous roles diagnosis, staging, screening and follow up. Several imaging modalities can be used to diagnose HCC including ultrasound, helical CT scanning, and
Ultrasound (US) plays a major role in the diagnosis of HCC, because it provides real-time and non-invasive observation by a simple and easy technique. In addition, US-guided needle puncture methods are frequently required for the diagnosis and/or treatment process of HCC. The development of digital technology has led to the detection of blood flow by color Doppler US,and the sensitivity for detecting tumor vascularity has shown remarkable improvement with the introduction of microbubble contrast agents. Moreover, near real- time 3-dimensional US images are now available. As for the treatment of HCC, high intensity focused ultrasound (HIFU) was developed as a novel technology that provides a transcutaneous ablation effect without needle puncture. The quality of a US examination is however operator dependent.
·
Sonographicaly the appearance of HCC· is variable. The masses are usually solid and may be hypoechoic , hyperechoic or of mixed echogenicity . These tumors more frequently invade portal or hepatic veins . Portal vein invasion is much more common than hepatic vein involvement,but hepatic vein involvement is more specific. Tumors invading the hepatic vein may extend to the IVC or the right atrium. Tumor invasion into the hepatic duct or common bile duct can be seen in 2-6% of advanced HCC’s. Obstructive jaundice can occur from direct invasion of the biliary tree or by compression by the tumor or lymph nodes.
·
Doppler sonography : HCC demonstrates internal vascularity (blood flow running into and branching within the tumor) and basket pattern of vascularity (a fine blood flow network surrounding the tumor nodule) .
·
Contrast Enhanced ultrasound :· On CEUS , the characteristic pattern of HCC is represented by an intense and fast peak of enhancement in the arterial phase , followed by a relatively quick washout staring at the beginning of the portal phase and quickly increasing towards the sinusoidal phase . Chaotic peritumoral and intralesional tortuous “corkscrew”/ “s” shaped vessels may be seen in he arterial phase as well as during the overlap with the portal phase.·Vascular lakes subsequently develop.
·
CT
·
Proper technical performance of CT with imaging in the hepatic arterial and portal venous phases, as well as delayed contrast images, is important in detecting hepatocellular carcinoma (HCC). Arterial phase imaging is most useful for the detection of HCC as its predominant blood supply is from the hepatic artery. However, it is less sensitive for the detection of small HCC and for dysplastic nodules which appear isodense to the liver parenchyma due to their predominant blood supply from the portal vein . CT arterio-portography and CT hepatic arteriography are more sensitive for the detection of HCC but the false positive rate is high due to benign hypervascular lesions like arterioportal shunts.Lesions may be missed if early vascular imaging is not performed. It is important to use high injection rates and appropriate bolus timing. Sensitivity of good-quality dual- or triple-phase CT for the detection of patients with tumors is 60-70%. The classical appearance of a HCC on a dynamic CT scan of the liver is that of early arterial phase enhancement followed by a rapid washout on a later phase .
- Ø Unenhanced CT typically reveals an iso-hypodense mass. If the mass is large, central areas of necrosis may be seen. Look for signs of cirrhosis or hemochromatosis.
- Ø In the hepatic arterial phase, lesions typically are hyperdense (relative to hepatic parenchyma) as a result of hepatic arterial supply. Larger tumors may have necrotic central regions that are typically hypodense during this imaging phase. Look for neovascularity to indicate the presence of inconspicuous lesions.
- Ø In the portal venous phase, small lesions may be isodense or hypodense and difficult to see, since the remainder of the liver increases in attenuation. Larger lesions with necrotic regions remain hypodense.
- Ø In the delayed-postcontrast phase, small lesions may be inconspicuous on late phases. Delayed phase scans may show a tumor capsule, one of the more specific signs indicating HCC.
CT can also evaluate complications of HCC such as portal venous or hepatic venous invasion. In addition, other complications·such as·bleeding within the tumor and hemoperitoneum can also be identified.
·
MRI
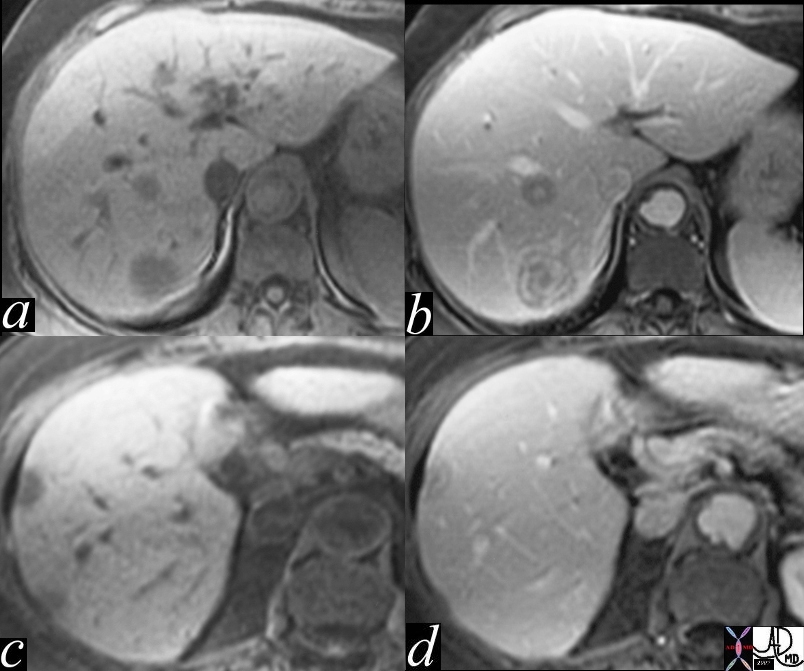
Magnetic resonance imaging (MRI) is extremely useful in the detection and characterization of regenerating and dysplastic nodules and HCC. The various techniques of MRI are useful and more sensitive than other modalities in the early detection and characterization of HCC in the cirrhotic liver and in differentiating it from dysplastic nodules and pseudolesions.
MR features of focal liver lesions in cirrhosis
|
Lesion |
T1 W image |
T2 W image |
Contrast enhancement pattern |
SPIO uptake |
Other features |
|
Regenerative nodule |
Variable |
Hypointense |
Enhances during portal venous phase |
Present |
Siderosis |
|
Dysplastic nodule |
Hyperintense |
Hypointense |
Enhances during portal venous phase |
Present |
Siderosis, nodule-in- nodule |
|
HCC (small) |
Hypointense |
Hyperintense |
Enhances during arterial phase |
Absent |
Nodule-in-nodule |
|
HCC (large) |
Heterogeneous |
Hyperintense |
Enhances during arterial phase |
Absent |
Fibrous capsule, satellite |
|
· |
· |
· |
· |
· |
nodules, invasion, fat |
|
Pseudolesion |
Variable |
Hypointense |
Enhances during arterial phase |
Absent |
· |
·
PET scan
PET scan has been evaluated in the experimental setting, but, to date, its role is uncertain. Routine use of PET scan for diagnosis or staging of hepatocellular carcinoma is not recommended.
·
Biopsy
Imaging studies such as triple-phase CT and contrast-enhanced MRI have evolved to the extent that biopsy of liver lesions is generally reserved for select ambiguous circumstances.
- Ø In general, core biopsy is favored over fine needle biopsy since larger amounts of tissue, often with normal surrounding parenchyma, can be obtained.
- Ø Controversy exists regarding the potential risk of tumor seeding along the needle tract. Some studies report a small increase in risk (approximately 1/1000), while others show no difference. Regardless, potential risks and complications should be considered before performing a biopsy.
- Ø Biopsy may be omitted in a clinical setting of a growing mass in a cirrhotic liver (>2 cm) noted on 2 coincident imaging techniques with at least one imaging showing contrast enhancement. Likewise, a growing mass in a cirrhotic liver on one imaging modality with an associated AFP level greater than 500-1000 ng/mL is clinically diagnostic of hepatocellular carcinoma. The need for biopsy should be carefully evaluated, especially if the risk for complications is high.
- Ø Biopsy is generally obtained percutaneously under ultrasonographic or CT guidance. Prior to obtaining biopsy, large-volume paracentesis may be useful in patients with massive ascites; similarly, platelet transfusion may be necessary in patients with cirrhosis with severe thrombocytopenia (<50,000). Bleeding risk does not correlate with elevations in prothrombin time.
- Ø Lesions that are 2-3 cm or smaller may be dysplastic nodules in a cirrhotic background. These are probably premalignant, and obtaining a biopsy is especially important to distinguish them from hepatocellular carcinoma. False-negative rates as high as 30-40% have been reported for biopsied tumors smaller than 2 cm in size.
- Ø Using laparoscopic guidance may make obtaining a percutaneous biopsy easier. Laparoscopy allows visualization of the liver to evaluate the extent of cirrhosis if surgery is being contemplated.
- Ø Obtaining a biopsy may be unnecessary in patients who will undergo resection regardless of diagnosis.
Staging
The goal of tumor staging is to separate patients into different groups based on their predicted survival to help determine the most appropriate treatment modality. The currently available staging systems for HCC include the pathologic tumor-node-metastasis (pTNM), Okuda, Cancer of the Liver Italian Program (CLIP), and Barcelona Clinic Liver Cancer.
- Ø TNM staging criteria for hepatocellular carcinoma
- T1 – Solitary tumor without vascular invasion
- T2 – Solitary tumor with vascular invasion or multiple tumors none more than 5 cm
- T3 – Multiple tumors more than 5 cm or tumor involving a major branch of the portal or hepatic vein(s)
- T4 – Tumor(s) with direct invasion of adjacent organs other than the gallbladder or with perforation of visceral peritoneum
- N0 – Indicates no nodal involvement
- N1 – Indicates regional nodal involvement
- M0 – Indicates no distant metastasis
- M1 – Indicates metastasis presence beyond the liver
- Ø Stage grouping
- Stage I = T1 + N0 + M0
- Stage II = T2 + N0 + M0
- Stage IIIA = T3 + N0 + M0
- Stage IIIB = T4 + N0 + M0
- Stage IIIC = TX + N1 + M0
- Stage IVB = TX + NX + M1
- Ø CLIP scoring system: Score of 0-2 is assigned for each of the 4 features listed below; cumulative score ranging from 0-6 is the CLIP score.
- Child-Pugh stage
- Stage A = 0
- Stage B = 1
- Stage C = 2
- Tumor morphology
- Uninodular and extension less than 50% = 0
- Multinodular and extension less than 50% = 1
- Massive and extension greater than 50% = 2
- Alpha-fetoprotein
- Less than 400 = 0
- Greater than 400 = 1
- Portal vein thrombosis
- Absent = 0
- Present = 1
Estimated survival based on CLIP score: Patients with a total CLIP score of 0 have an estimated survival of 31 months; those with score of 1, about 27 months; score of 2, 13 months; score of 3, 8 months; and scores 4-6, approximately 2 months.
·
·
Management
Treatment approach to patients with hepatocellular carcinoma. The initial clinical evaluation is aimed at assessing the extent of the tumor and the underlying functional compromise of the liver by cirrhosis. Patients are classified as having resectable disease, unresectable disease, or as transplantation candidates. Abbreviations: OLTX, orthotopic liver transplantation; TACE, transarterial chemoembolization; PEI, percutaneous ethanol injection; RFA, radiofrequency ablation; LN, lymph node. Child’s A/B/C refers to the Child-Pugh classification of liver failure.
·
Most HCC patients have two liver diseases, cirrhosis and HCC, each of which is an independent cause of death. The presence of cirrhosis usually places constraints on resection surgery, ablative therapies, and chemotherapy. Thus patient assessment and treatment planning have to take the severity of the nonmalignant liver disease into account. A multidisciplinary team, including a hepatologist, interventional radiologist, surgical oncologist, transplant surgeon, and medical oncologist, is important for the comprehensive management of HCC patients.
·
·
Prognosis
The natural history of HCC is highly variable. Patients presenting with advanced tumors (vascular invasion, symptoms, extrahepatic spread) have a median survival of ~4 months, with or without treatment.
Conclusion
HCC is an aggressive tumor with a high case fatality rate. Successful treatment is dependent on early detection through widespread use of surveillance of patients at risk for HCC development.
Cholangiocarcinoma
·
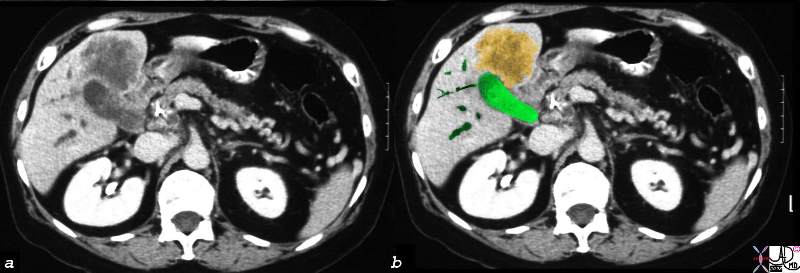
16254c02b.8s |
| 16254c02b.8s gallbladder anterior wall liver invasion space occupatopn obstruction bile ducts aggressive gallbladder carcinoma complicated by direct invasion metastasis liver windows narroe windws tumor settings gallbladder fossa GBF CTscan Courtesy Ashley Davidoff copyright 2008 |
·
18126 |
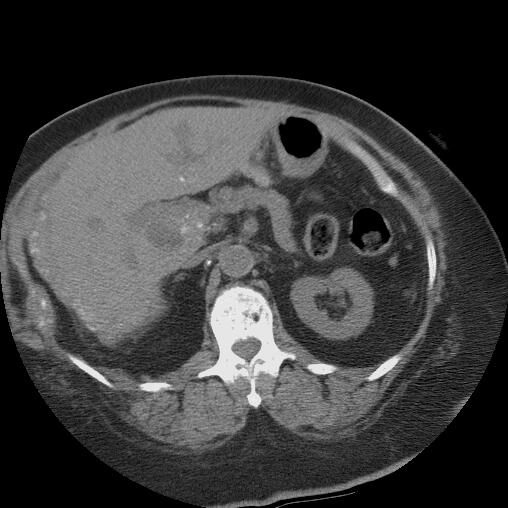
| 18126 liver + fx mass + fx calcification + bile duct + cholangiocarcinoma + imaging radiology CTscan splenic early heterogeneous contrast enhancement |
Hepatoblastoma
Hepatoblastoma is rthe most common malignant tumor of the liver in the pediatric population
·
·

03347c03.8s |
| 03347c03.8s 18 month old female presents with abdominal distension and lethargy, and clinica lexam shows an enlarged liver. The grosspathology specimen shows white fibrous bands (f) mixed with dark pink tumor (t) and pockets of marroon hemorrhage(h). liver malignancy hepatoblastoma heterogeneous mass grosspathology Courtesy Ashley Davidoff MD copyright 2009 all rights reserved |

03334.81s |
| 03334.81s liver hepatic malignancy hepatoblastoma heterogeneous mass hemorrhage necrosis fibrous scar grosspathology Courtesy Ashley Davidoff MD copyright 2009 all rights reserved |

03347c02.8s |
| 03347c02.8s 18 month old female presents with abdominal distension and lethargy, and clinica lexam shows an enlarged liver. The histopathology shows fibrous bands in light pink (f) mixed with cellular tumor (t). liver malignancy hepatoblastoma heterogeneous mass histopathology Courtesy Ashley Davidoff MD copyright 2009 all rights reserved |
·

03347c07.8s |
| 03347c07.8s 18 month old female presents with abdominal distension and lethargy, and clinical exam shows an enlarged liver. The histopathology shows advancing malignant cells and tumor (t in image b) overlaid in pale green, and intermediate zone (i) that has a pushing edge of malignant cells and a larger front of squashed normal liver cells (pseudocapsule) and then beyond and to the right of the image overlaid in red is normal liver (liver). liver malignancy hepatoblastoma heterogeneous mass histopathology space ocupation pressure atrophy pseudocapsule Courtesy Ashley Davidoff MD copyright 2009 all rights reserved |
Malignant Hemangioendothelioma
·
16700c001 |
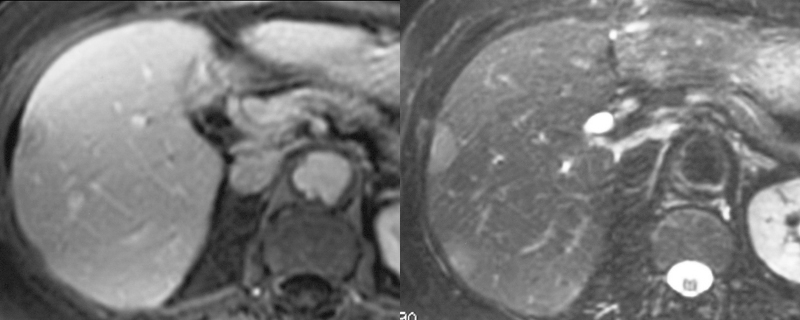
| 16700c001 liver hepatic nodules masses ring enhancement flattened lesion dimple on liver capsule multiple adult malignant hemangioendothelioma T1 weighted with and without gadolinium contrast MRI T1 weighted pre and post contrast Davidoff MD |
·
43346 |
| 43346 hx 45M with abdominal mass liver fx situs inversus fx stomach fx displaced fx large exophytic mass dx leiomyosarcoma situs inversus CTscan Courtesy Ashley Davidoff MD |
·
20921 |
| 20921 spleen + fx dense hyperdense parenchyma dx Thorotrast + imaging radiology CTscan C- non contrast liver dense hyperdense parenchyma calcific density calcification calcified infiltrative disease retciculoendothelial system RES |
15762 |
| 15762 Courtesy Ashley Davidoff MD code liver metastasis breast dx carcinoma hepar lobatum imaging radiology CTscan DB |
| 44408 |
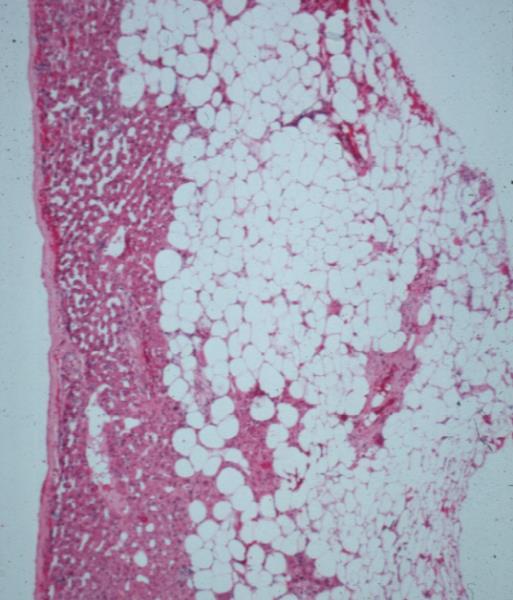
| 44408 liver applied biology capsule angiomyolipoma histopathology Courtesy Barbara Banner |
·
| 16700c02 |
| 16700c02 liver hepatic nodules masses ring enhancement flattened lesion dimple on liver capsule multiple adult malignant hemangioendothelioma T1 weighted with and without gadolinium contrast MRI T1 weighted pre and post contrast Davidoff MD |
·
·
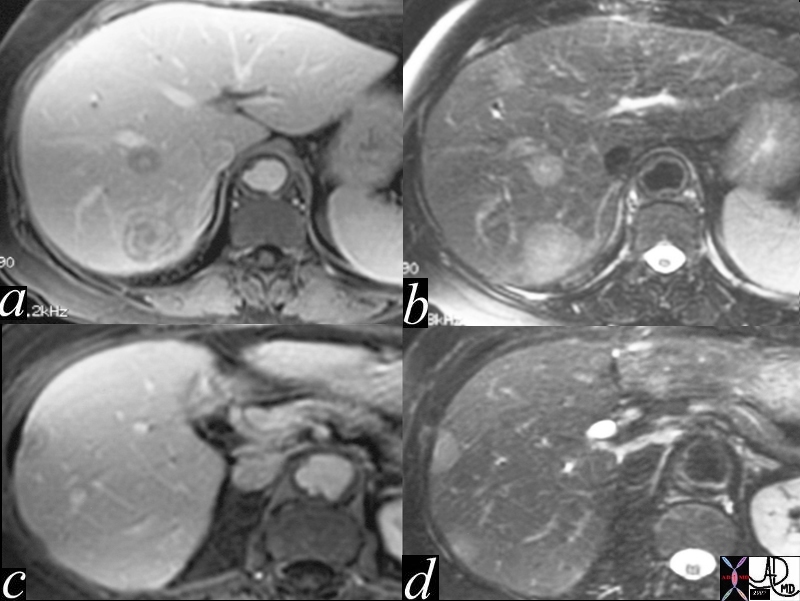
16700c004 |
| 16700c004 liver hepatic nodules masses ring enhancement flattened lesion dimple on liver capsule T2 bright multiple adult malignant hemangioendothelioma T1 weighted with gadolinium contrast MRI T1 weighted post contrast T2 ewighted Davidoff MD |
·
·
Metastases
·
·
|
The Battleground – Tumor vs Healthy Tissue |
|
The histopathology of a surgically resected hepatic metastasis from a primary colon carcinoma shows a mucinous secreting tumor at the bottom of the image labeled “advancing tumor”. The cellular detail is not well seen in the low power view but the columnar epithelial nature, hyperchromicity, and bizarre appearance of squatter tissue in the liver are recognized as malignant characteristics. The white spaces in between the cellular structure, represents mucin in the tumor. The edge of the tumor shows a rounded and aggressive push into the liver where the injured liver in the battlefield is recognized as compressed normal liver with linear areas of hemorrhage·evidenced by a curvilinear line at 11 and 12 oclock of· increased area of redness representing ·extravascular red cells (“layer of “injured liver”). The uppermost layer represents the normal liver which at this time looks healthy but is in jeopardy. The radial red spokes seen at the edge of the tumor are artifacts of preparation but are indicators of the direction of the battle. ·liver cancer metastasis malignant tumor battlefield mucinous secreting adenocarcinoma colon primary histopathology destruction hemorrhageCourtesy Ashley Davidoff MD copyright 2009 all rights reserved 5star 03028b01.81s |
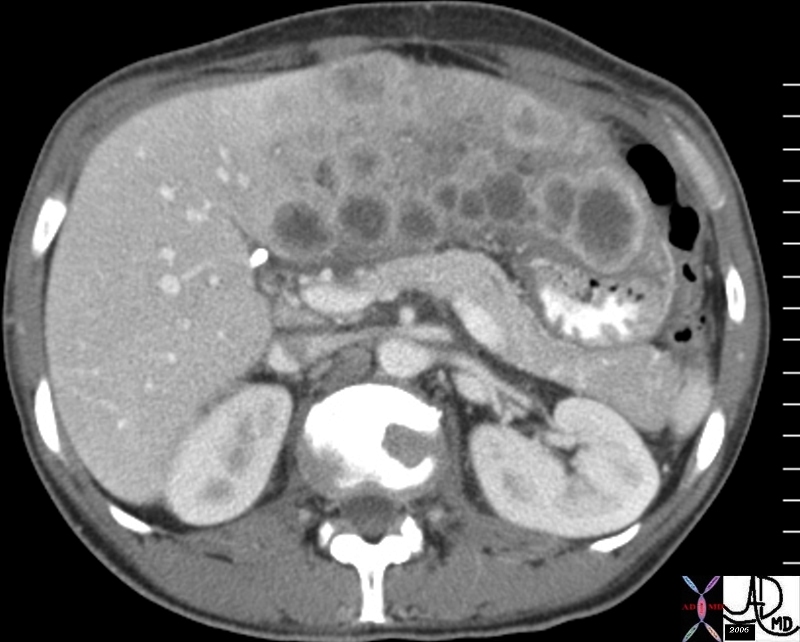
| 46587.800 |
| 46587.800 liver bone vertebra fx mass space occupying disease dx liver metastases metastasis dx primary lung carcinoma malignancy cancer CTscan Davidoff MD |
·
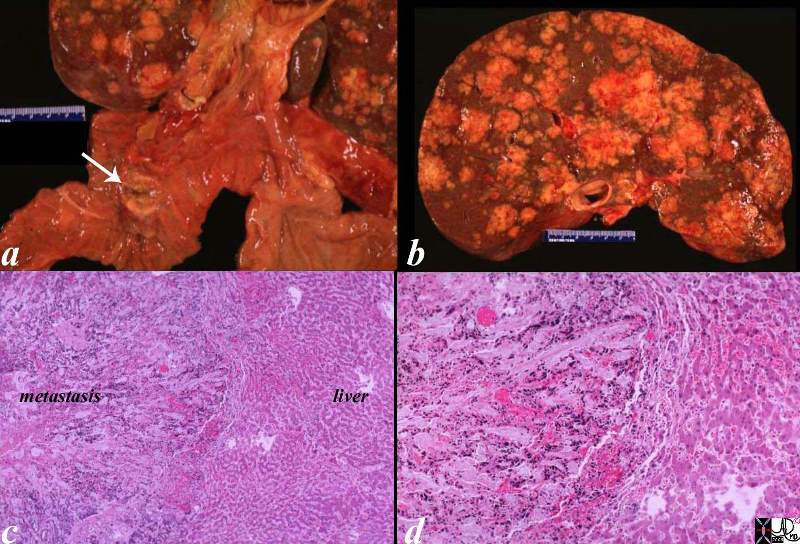
02643c03.8s |
| 02643c03.8s The post mortem specimen (a) shows a 2cms primary duodenal carcinoma (white arrow) with extensive nodular hepatic metastases occupying 80% of the liver. (b) In image c, the metastasis reflects an overall a blue hue caused by the increased nuclear presence. The image reveals an irregular and advancing threat on the normal liver. The higher power in d, shows a rind of compressed liver that is in the process of destruction and hemorrhage (red patches) in the tumor, in the border, as well as in the normal liver. This image is reminiscent of an advancing enemy in a battlefield with victory apparent for the aggressor. code liver duodenum adenocarcinoma metastases metastasis cancer malignant space occupation pressure atrophy death grosspathology histopathology Courtesy Ashley DAvidoff MD copyright 2009 all rights reserved |
| 40343c |
| 40343c Courtesy Ashley Davidoff MD code pancreas pancreatic body fx mass code liver fx multiple nodules and masses code dx primary adenocarcinoma of the pancrreas complicated by metastasis metastatic disease to the liver imaging radiology CTscan neoplasm malignant tumor cancer |
·
|
Normal Liver –·(left) ·and Cancer in the Liver (right) |
|
The liver on the left is normal except for gallstones in the gallbladder.· Gallstones are squatters but they usually do not have aggressive tendencies unless they block the outflow of bile.· The liver shown in the right image is from a patient who succumbed to metastatic pancreatic carcinoma.· The normal liver cells have been displaced and have been overridden by metastatic squatters.· The metastases have contributed to the demise of the patient by their relentless displacement of normal tissue, causing progressive impairment of function.· 13456 liver normal anatomy gallbladder stones cholelithiasis grosspathology 13448b01 |
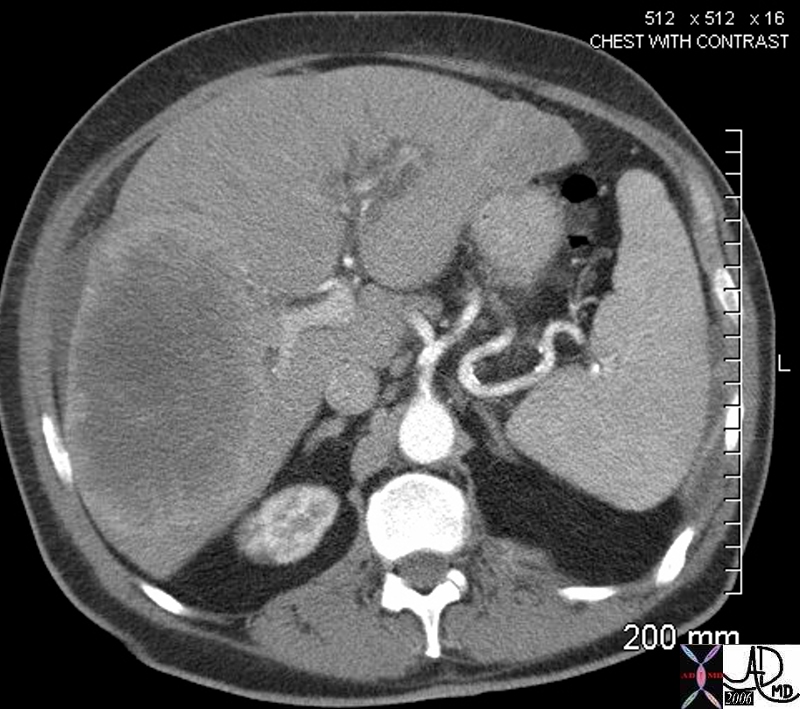
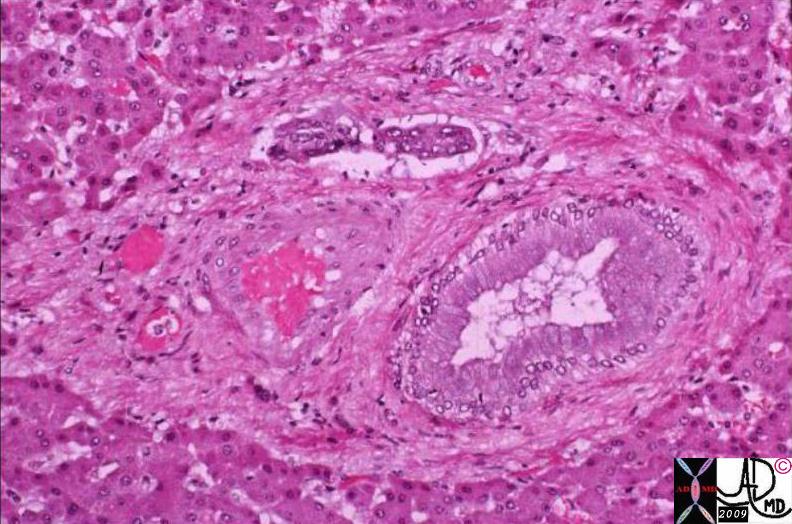
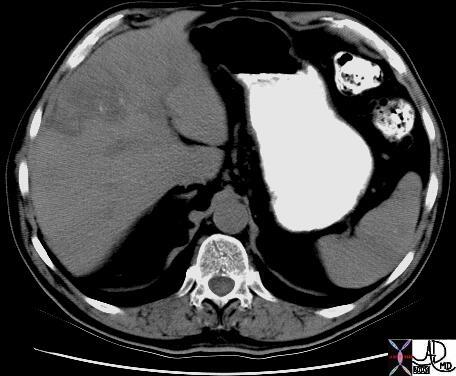
 02891c03.8s 02891c03.8s |
| 02891c03.8s This unfortunate patient presented in pregnancy with metastatic disease to the liver showing rounded discrete metastases in the liver on CTscan (a) confirmed at autopsy to be metastatic pancreatic carcinoma. The lesions in the liver (b) show a bile tinged green halo caused by local cholestasis caused by space occupation and obstruction. In c and d, the metastatic tumor (m) is seen to the left of the images, the middle layer of bile stained injured liver cells caused by cholestsais (ch) is seen as a subtle orange tinged section (c). The bile tinged layer (ch) is digitally enhanced in orange in d. Therelatively normal liver tissue (l) is seen to the right and is enhanced in with pink (d). code liver malignant cancer pancreatic primary pancreas bile tinge cholestasis space occupation bile stain CTscan grosspathology histopathology Courtesy Ashley Davidoff MD copyright 2009 all rights reserved |
 45220 45220 |
| 45220 liver fx large hepatic mass dx colonic rectosigmoid carcinoma with hepatic metatsasis s/p stent placement CTscan Courtesy Ashley Davidoff MD 45220 45221 45222 |
| ·
|
| These two cases represent the microscopic events described in the macroscopic examples above.· The left image shows normal liver cords at high power, while the image on the right shows slightly lower power of the liver cords in pink, and a blue, rounded, space occupying metastasis. ··
· 13445 11440 liver hepatocytes normal metastasis adenocarcinoma histopathology nodule Davidoff MD |
 02986.8s 02986.8s |
| 02986.8s This specimen is from a biopsy of the liver. The medium power view of malignant adenocarcinoma shows groups of columnar cells arranged in rosettes in a bed aof malignant stromal tissue and surrounded by normal liver. Although the nuclii in this case are not particulalrly enlarged the cytoplasm is basophilic, and the appearance is of a “squatter” in the liver, ie tissue that does not belong, is consistent with metastatic adenocarcinoma malignant cancer liver hepatic adenocarcinoma histopathology Courtesy Barbara Banner MD copyright 2009 all rights reserved GW question 5star |
 18370 18370 |
| 18370 liver colon large bowel liver fx hepatic mass fx fine stippled calcifications calcified mass dx metastattic mucin secreting adenocarcinoma of colon carcinoma metastasis CTscan Courtesy Ashley Davidoff MD |
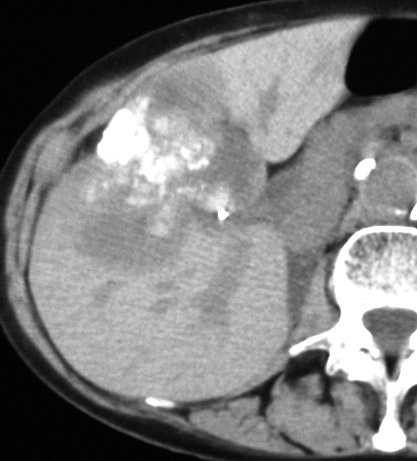 39484 39484 |
| 39484 liver CTscan imaging radiology mass dystrophic calcification calcified neoplasm mass metastasis malignant carcinoma adenocarcinoma cancer tumor |
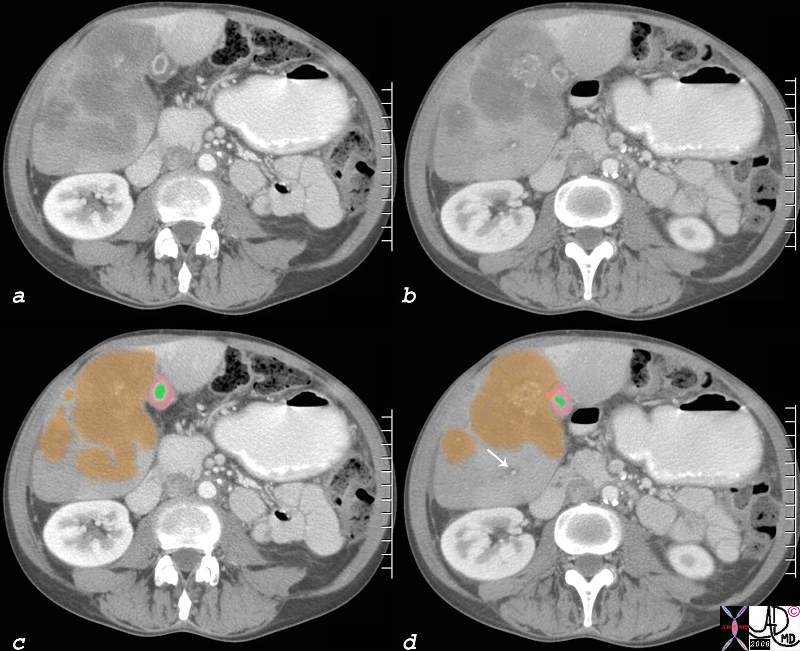 82313c01.8s 82313c01.8s |
| 82313c01.8s 51 year old male with history of rectal carcinoma with extensive hepatic metastases. (orange) The metastases surround gallbladder and by space occupation result in venous and lymphatic congestion causing a thickened edematous gallbladder wall (orange) with a small lumen (green) Tthe portal triad shows surrounding edema (white arrow). This is called periportal tracking which is another sign of venous and lymphatic engorgement. The metastases are calcified confirming the mucinous nature of the tumor. calcified metastasis in gallbladder fossa edema of the wall lymphatics contracted gallbladder CTscan copyright 2008 Courtesy Ashley Davidoff MD gallbladder liver portal triad |
 03034c.8s 03034c.8s |
| 03034c.8s The gross and histopathological specimens are from the autopsy of a patient who died of breast carcinoma that was metastatic to the liver. The right lobe of the liver shows two patterns of metastases. In the posterior aspect of the liver there is diffuse disease, and in the anterior aspect there are innumerable nodules of varying sizes. The left lobe is relatively spared, and the pattern is of a nodular nature. The low power histopathological section shows a diffuse infiltrate of hyperchromatic malignant nuclii in the bottom of the image and relatively normal liver in the upper portion. code liver metastases metastasis breast primary cancer malignant space occupation infiltration infiltrative pattern right lobe left lobe grosspathology histopathology Courtesy Ashley DAvidoff MD copyright 2009 all rights reserved GW question |

02939c01.8s |
| 02939c01.8s The series is from a patient who died of metastatic adenocarcinoma. In image a, the ultrasound shows diffuse increased echogenicity in the dome of the right lobe of the liver that suggests diffuse infiltration of metastatic disease. the liver at autopsy showed a combination of diffuse infiltration and discrete nodules. It seems that the discrete nodules in this case become confluent to become infiltrative disease. The histolopathology in d shows discrete metastatic deposits of glandular type metastases separated by spaces within the lesion of mucus consistent with metastatic mucinous adenocarcinoma in a bed of malignant stroma. The distinct blueness and “badness of the malignant tissue is reflective of the hyperchromicity of the nuclii and their dominance in the cell. code liver metastases metastasis discrete diffuse ultrasound grosspathology histopathology Courtesy Ashley Davidoff MD copyright 2009 all rights reserved GW question |
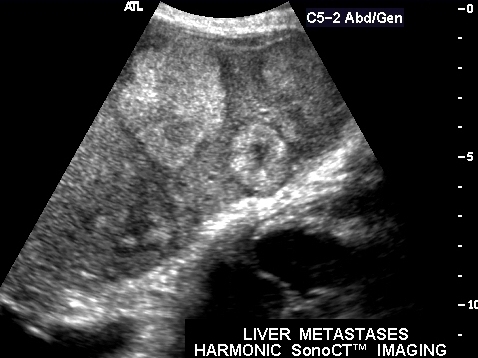
739473 |
| 39473 Courtesy Philips Medical Systems liver imaging radiology USscan |

24012c01.8s |
| 24012c01.8s The ultrasound of the liver is from a 72 year old patient with metastatic breast carcinoma. There is a region overlaid in green ) that is hyperechoic and diffuse without clear nor rounded borders. No obvious mass or nodule can be defined. This finding represents the less common infiltrating metastatic pattern characteristically, but not exclusively observed in patients with breast carcinoma. code liver cancer malignancy malignant carcinoma metastasis infiltrating infiltrative US USscan ultrasound Courtsy Ashley DAvidoff MD copyright 2009 all rights reserved |
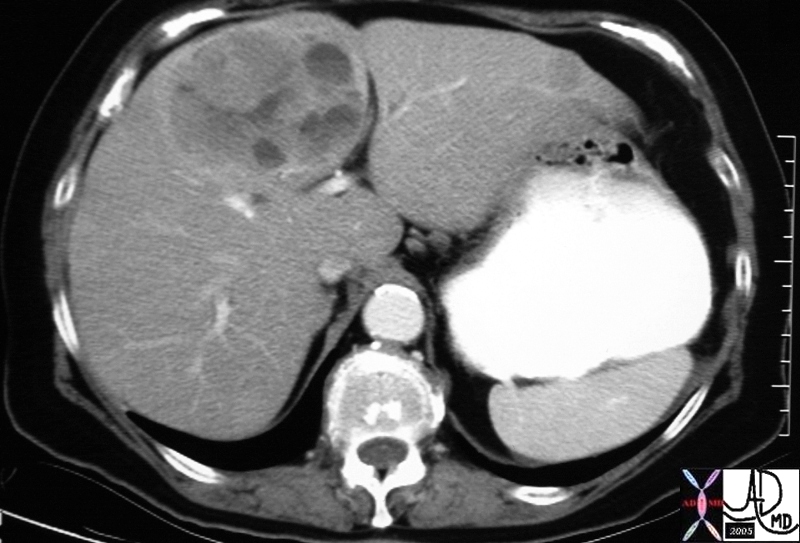
20912 |
| 20912 liver + fx mass + complex cystic solid metastasis dx carcinoid tumor + imaging radiology CTscan C- mass neoplasm malignant metastasis carcinoma cancer tumor Courtesy Ashley Davidoff MD |

02996c02.8s |
| 02996c02.8s The autopsy series is from an elderly female who died with metastatic ovarian carcinoma. The CTscan (a) shows a focal hypodensity at the edge of the right lobe of the liver that was shown to be a metastasis. There is ascites around the spleen. Image b shows a white patch on the liver capsule in the region of the CT finding. Image c shows metastatic disease on the diaphragm characterized by a white rind of malignant tissue. (orange arrow). The histipathology shows three layers of tissue with normal liver at the bottom, the capsule in the middle and the matastatic deposit on the edge of the capsule. liver capsule ovary metastasis malignant primary cancer metastases transpertoneal spread of disease diaphragm Courtesy Ashley Davidoff MD copyright 2009 all rights reserved GW question |

24776 R W c |
| 24776 R W c liver + lymph nodes lymphatics+ lymphatic drainage portal triad + fx normal + anatomy + imaging radiology CTscan |
·
20115c |
| 20115c Courtesy Ashley Davidoff MD code pancreas + dx carcinoma code abdomen fx abdominal ascites spleen stomach liver gastrolienal ligament lesser omentum gastrohepatic ligament masses metastasis abdomen + imaging radiology CTscan primary pancreas neoplasm malignant carcinoma cancer tumor |
·
22993 |
| 22993 liver + capsule + spleen fx mass nodule + fx calcification + calcified dx pseudomyxoma peritonei + imaging radiology CTscan |
·

03146c.8s |
| 03146c.8s 56 year old male with gastric carcinoma and T2 weighted MRI that shows diffuse infiltration of metastic disease (areas of increased water (white) are abnormal and the darker areas are normal liver. The liver is almost totally replaced by diffuse metastases. The histopathology shows an infiltrative patternof dark tissue caused by hyperchromatic nuclii and diminished basophilic cytoplasm. The pink cells in between represent the normal liver. code liver metastases metastasis stomach primary gastric metastases to liver hepatic infiltrative malignant malignancy cancer MRI T 2 weighted image histopathology Courtesy Ashley DAvidoff MD copyright 2009 all rights reserved |
·
22359 |
| 22359 51 M hepatic bare area liver abdomen ascites fx mass fx stippled calcification calcified fx psammomatous psamomatous calcifications dx metastattic mucinous adenocarcinoma of the rectum CTscan soft tissue windows Davidoff MD 22359 22357.800 |
·
·
16868 |
| 16868 diaphragm liver porta hepatis gastrohepatic ligament Glissons capsule + fx calcification + primary ovary + metastasis + imaging radiology CTscan neoplasm malignant carcinoma tumor cancer |
·
·
20893 |
| 20893 spleen + capsule + fx calcification + metastatic primary ovary + carcinoma + imaging radiology CTscan C- liver Glisson’s capsule ligamentum venosum + fx calcification falciform ligament ligamentum teres fx calcium calcified dx metastatic primary ovary neoplasm malignant metastasis metastases cancer tumor abdomen peritoneum peritoneal cavity calcium deposit calcification calcified punctate ascites neoplasm malignant metastasis metastases cancer tumor |
03779.800 |
| 03779.800 liver metatstasis primary ovarian cancer adenocarcinoma psammomatous calcifications psammoma bodies glandular malignancy histopathology Davidoff MD |
·
20886 |
| 20886 code spleen + capsule + fx calcification + metastatic primary ovary + carcinoma + imaging radiology CTscan C- liver Glisson’s capsule + fx calcification + metastatic primary ovary gastrohepatic gastrolienal falciform ligament ligamentum teres fx calcium calcification dx metastatic primary ovary neoplasm malignant metastasis metastases cancer tumor |
·
03780 |
| 03780 liver + fx psammomatous calcification + dystrophic calcification calcium metastatic primary ovary ovarian carcinoma + cytopathology |
·
03028c01 |
| 03028c01 The images show a series of 4 images that reveal a metastaic lesion in the liver from a primary colon carcinoma. The lesion is about 2.5cms in size, has rounded pseudopod like advancing edges (best seen in image b – CT and image C MRI), is situated in the superior aspect of the right lobe of the liver and appears to have a complex nature. The through transmission seen in the ultrasound (a), the low density seen in the CT(b) and brightness on T2 (d) suggest complex fluid. The bright central region and bright lobulated rim on T1 (c) suggest mucin and or hemorrhage. liver hepatic mass a= US through transmission b= CT hypodense lesion c = T1 weighted cental intensity rim and matrix of low intensity periphery hyperintense d = T2 weighting = T2 bright fx metastattic colon carcinoma mucin secreting adenocarcinoma with central hemorrhage peripheral hemorrhage USscan CTscan MRIscan Davidoff MD copyright 2009 all rights reserved |
·
03028c02 |
| 03028c02 The images show a series of 4 images that reveal a metastaic lesion in the liver from a primary colon carcinoma. The lesion is about 2.5cms in size, has rounded pseudopod like advancing edges (best seen in image a – T1 weighted MRI), is situated in the superior aspect of the right lobe of the liver and appears to have a complex nature. The brightness on T2 weighted series (d) and white area in the histopathology (d) suggest water content made up from mucus. The bright central region and bright lobulated rim on T1 (a) suggest mucin and or hemorrhage. The gross pathology shows central necrosis with hemorrhage (c) and the histopathology shows mucinous tumor with hemorrhage at the the advancing edge. liver hepatic mass a= MRI T1 weighted – T1 weighted cental intensity rim and matrix of low intensity periphery hyperintense b= MRI T2 weighted lesion is wet c = gross pathology shows central necrosis and hemorrhage and peripheral hemnorrhage d = histopathology advancing mucin secreting tumor with destroyed liver and hemorrhage fx metastattic colon carcinoma mucin secreting adenocarcinoma with central hemorrhage peripheral hemorrhage MRIscan liver hepatic mass histopathology grosspathology Courtesy Davidoff MD all rights reserved copyright 2009 |
·
·
16844 |
| 16844 Courtesy Ashley DAvidoff MD code pancreas + fx mass endocrine tumor + code liver capsule fx mass dx metastasis code IVC fx stent dx obstruction Budd Chiari imaging radiology CTscan neoplasm tumor carcinoma cancer |
| 46579 |
| 46579 liver hepatic vein fx tumor thrombus lung fx consolidation dx infiltrative adenocarcinoma of the lung with metastases to the liver and adrenal and invasion of the hepatic veins malignancy cancer Davidoff MD 46581 46588 46587 |
·
·
03057c01.8s |
| 03057c01.8s The collage consists of a CT scan (a) showing a focal well circumscribed low density liver lesion. Biopsy of this lesion yielded the histopathological specimen (b) that shows hyperchromatic nuclii and spindle shaped cells consistent with metastatic leiomyosarcoma. The cytological aspirate (c) shows spindle shaped nuclii with pleomorphism and hyperchromicity. code malignant cancer sarcoma histopathology CTscan Courtesy Ashley Davidoff MD copyright 2009 all rights reserved |
| 22068 |
| 22068 liver focal filling defect lymph nodes retrocrural enlarged dx lymphoma CTscan imaging radiology periportal tracking |
·
·
03172c01.8s |
| 03172c01.8s This series is from a patient who died with diffuse histiocytic lymphoma and DIC. The CT scan (a) shows hepatosplenomegaly with a low density liver suggestive of steatosis. The grosspathology specimen shows an enlarged liver with punctate hemorrhages. The low power histopathology shows a diffuse inflitration of dark nuclii reminiscent of a malignant infiltration, and the high power shows fatty vacuoles within the cytoplasm of the hepatocytes and an infiltrate of histiocytic appearing cells with increased nuclear cytoplasmic ratio (d). liver fatty change steatosis malignant cancer histiocytic lymphoma DIC metastases metastasis CTscan grosspathology histopathology Courtesy Ashley Davidoff MD copyright 2009 all rights reserved GW question |
·
| 70248c01 |
| 70248c01 liver metastattic small lung carcinoma with diffuse metatstattic disease to the liver )hepatic metastases metastasis before and after treatment 6weeks post chemotherapy successful result size change character change CTscan Davidoff MD 5star |
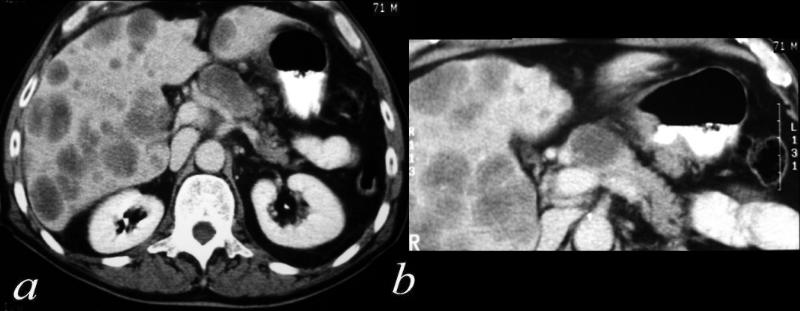
Carcinoma – Extending into the Liver via the Gallbladder Fossa
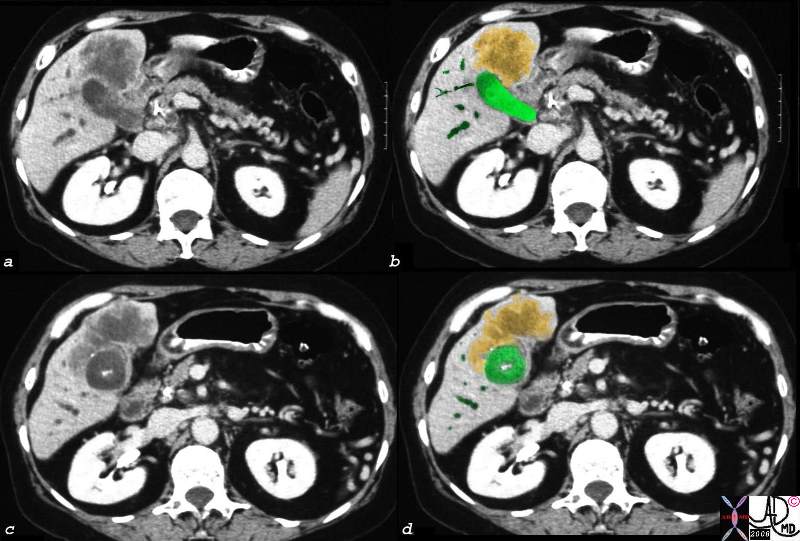
Direct Invasion into the Liver and Bile Duct Obstruction |
|
The CTscan of this patient shows a normal sized gallbladder (green) associated with a 4-5cms mass (orange) adjacent to the gallbladder, and extending from the gallbladder fossa. There is asssociated biliary obstruction (dark green tubes)· This case represents invasive gallbladder carcinoma with bile duct obstruction. Images c and d show the almost universal association of gallstones in patients with carcinoma.···In this case the stones (white) are in the centre of the gallbladder. (green) ·16254c03.8s gallbladder anterior wall liver invasion space occupatopn obstruction bile ducts aggressive gallbladder carcinoma complicated by direct invasion metastasis liver windows narroe windws tumor settings gallbladder fossa GBF CTscan Courtesy Ashley Davidoff copyright 2008 |
·
·
Dystrophic Calcification – Carcinoma of the Gallbladder Extending into the Gallbladder Fossa |
|
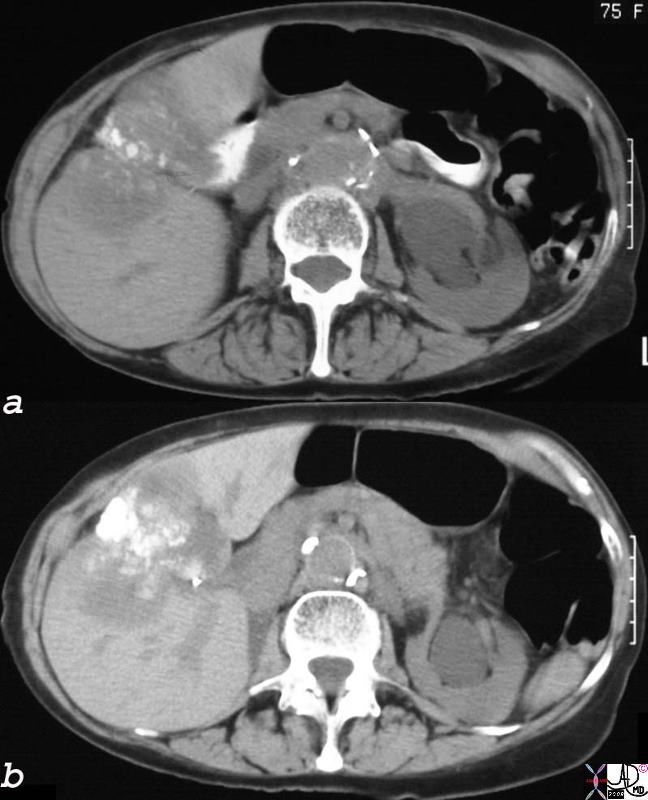
The non-contrast CT of the 75year old female patient, shows dystrophic calcification·in a mass that seems to be part of the wall of the gallbladder.· The·mass extends into the liver.· These findings are consistent with an inoperable gallbladder mucinous adenocarcinoma, metastatic to the liver by direct extension.· Calcification of adenocarcinomas is usually found in the mucinous variant of the disease. 24404c.8s 75 female gallbladder calcification adjacent mass in the liver local invasion into the gallbladder fossa dystrophic calcification probably mucinous adenocarcinoma of the gallbladder carcinoma stones cholelithiasis hydronephrosis |
·
Carcinoma – Extending into the Liver via the Gallbladder Fossa and Additional Metastases
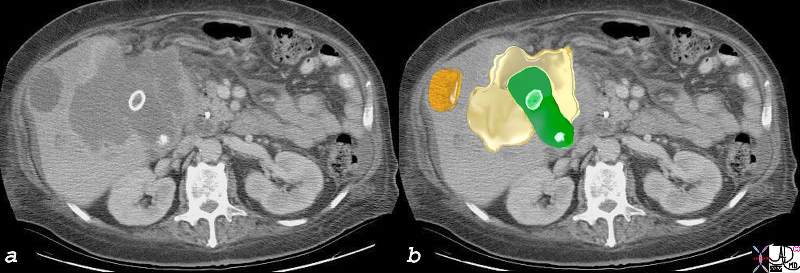
Local Invasion and Separate Liver Mestastasis |
|
The CTscan in this patient shows two calcified gallstones in the gallbladder (green), with a mass like malignant abnormality extending into the liver on either side (yellow) and a remote metastasis (orange) which is deforming the liver edge. 17280c02b01.8s gallbladder liver mass local invasion cholelithiasis metastasis carcinoma primary gallbladder gallbladder fossa Courtesy Ashley Davidoff MD copyright 2008 |
Disease Extension Beyond the Liver·
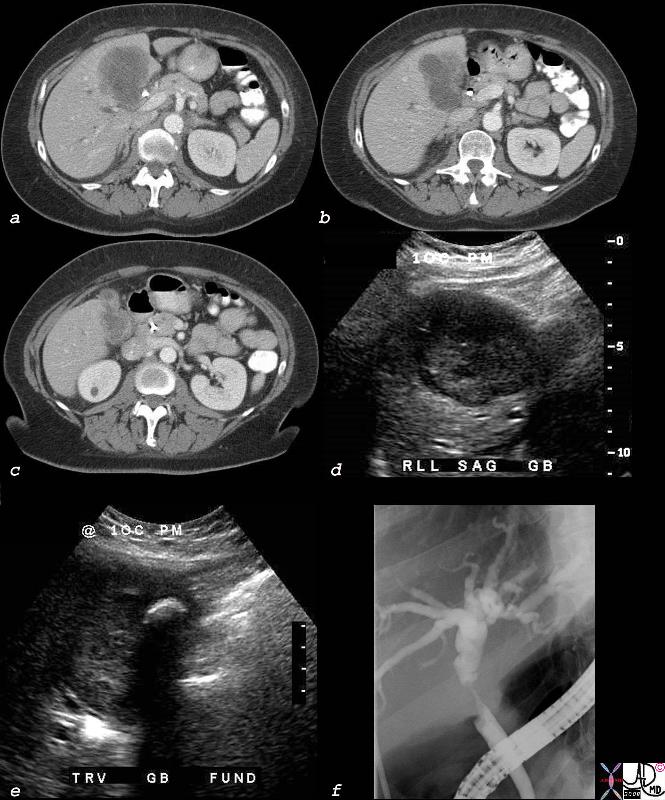
Cholangiocarcinoma with Involvement of the Gallbladder and Bile Duct |
|
The multiple imaging modalities of this patient who is elderly female who presents with obstructive jaundice.··Deformity of the gallbladder by CT is noted, with extension of a mass into the surrounding liver (a,b,c) In addition to a mass in the gallbladder (d), the ultrasound shows a shadowing stone (e).· An ERCP (f), shows a circumferential stricture of the bile duct.·The stricture is in the region of the cystic duct – bile duct junction, with non visualization of the cystic duct suggesting extension of the gallbladder cancer into the cystic duct and bile duct.· Lymph node involvement with bile duct obstruction is a less likely possibility due to the circumferential nature of the stricture. 40018c02b.8s elderly female cholelithiasis obstructive jaundice common bile duct stricture dilated intrahepatic ducts irregular enhancing thickening of the gallbladder wall stent gallbladder carcinoma with extension into the CBD or CBD carcinoma with extension into the wall of the gallbladder CTscan USscan ultrasound ERCP Courtesy Ashley DAvidoff MD copyright 2008 |
·
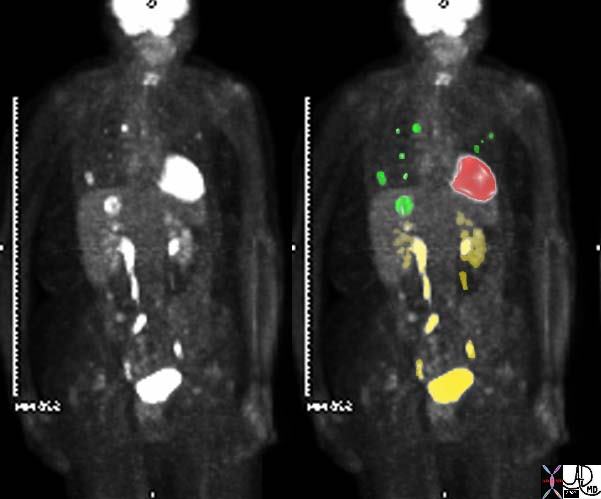
·
| 45132c01 |
| 45132c01 elderly female with known sigmoid colon carcinoma liver lungs mediastinum fx FDG positive uptake fx high intensity uptake in a single focus in the dome of the right lobe of the hepatic parenchyma meadiastinal lymph nodes and within the pulmonary parenchyma dx colonic mucinous adenocarcinoma complicated by metastatic lung disease and hepatic metastasis PETscan Courtesy Ashley Davidoff MD 45132 45133 45134 45135 45136 45139 45141 |
Red Flags
- ü Decompensation in patients with compensated cirrhosis
- ü The triad of abdominal pain, weight loss and an abdominal mass
- ü The presence of a nodule larger than 2 cm with hypervascular characteristics detected by imaging techniques
- ü Gross Vascular invasion is a marker of widespread disease.
References
- 1. Naugler WE and Schwartz JM. Hepatocellular carcinoma . Disease- a-hepatoblastomamonth. 2008 (54); 432-44.
- 2. Leong TYM and Leong ASY. Epidemiology and carcinogenesis of hepatocellular carcinoma . HPB 2005; 7:5-15.
- 3. Digumarthy SR, Sahani DV, Saini S . MRI in detection of hepatocellular carcinoma . Cancer Imaging. 2005; 5: 20-24.
- 4. Schafer DF and Sorrell MF. Hepatocellular carcinoma . Lancet 1999;353: 1253-57
- 5. http://www.guideline.gov/summary/summary.aspx?ss=15&doc_id=9401&nbr=5036
- 6. http://books.google.com/books?id=OoAkdMgCxV0C&pg=PA91&lpg=PA91&dq=hcc+causes+bile+obstruction&source=bl&ots=h_BDTaU4aB&sig=Mg64P-p7lgJu9TvpvfnHoI2ugpw&hl=en&ei=GKthSumdGYPYtgOi36Fn&sa=X&oi=book_result&ct=result&resnum=9
- 7. http://online.statref.com.ezproxy.library.tufts.edu/Document/Document.aspx?docAddress=Z-lA2-alKAgqyrbNXcUh1w%3d%3d&offset=6130&SessionId=FAC669MXIJSTVNEK
·
·
·
| 03347c07.8s |
| 03347c07.8s 18 month old female presents with abdominal distension and lethargy, and clinica lexam shows an enlarged liver. The histopathology shows advancing malignant cells and tumor (t in b) overlaid in pale green, and intermediate zonebands in light pink (f) mixed with cellular tumor (t). liver malignancy hepatoblastoma heterogeneous mass histopathology Courtesy Ashley Davidoff MD copyright 2009 all rights reserved |
The intimate relationship between the liver cells and the sinusoids infers that seedlings of malignant cells will aslo be in close proximity to the venous system and if they break off from the mother tumor have access to the rest of the body and the lungs in particular from the primary tumor
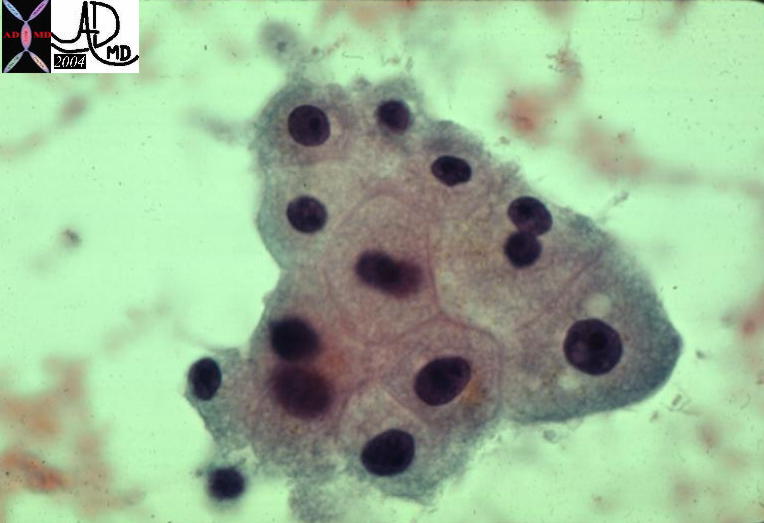
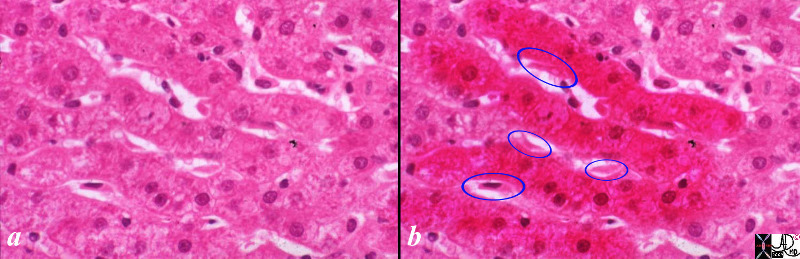
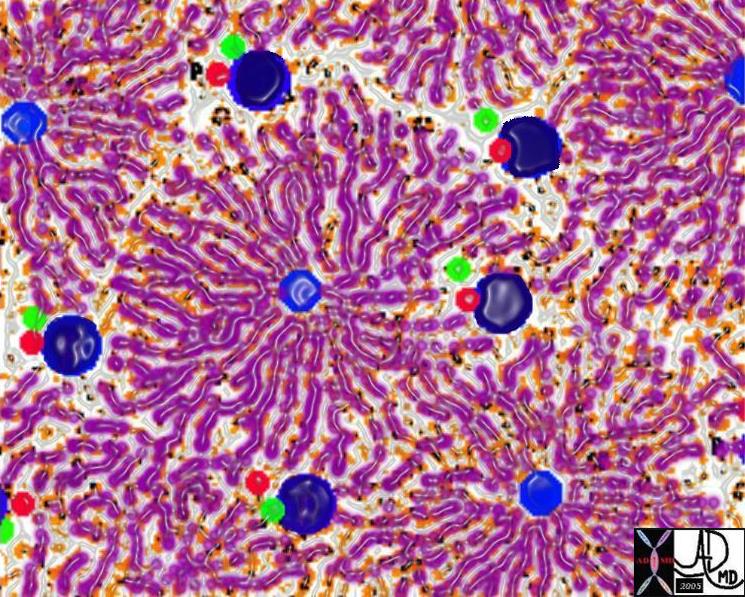



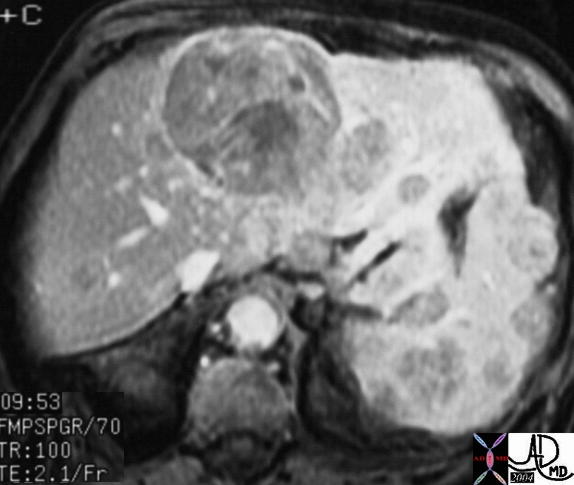
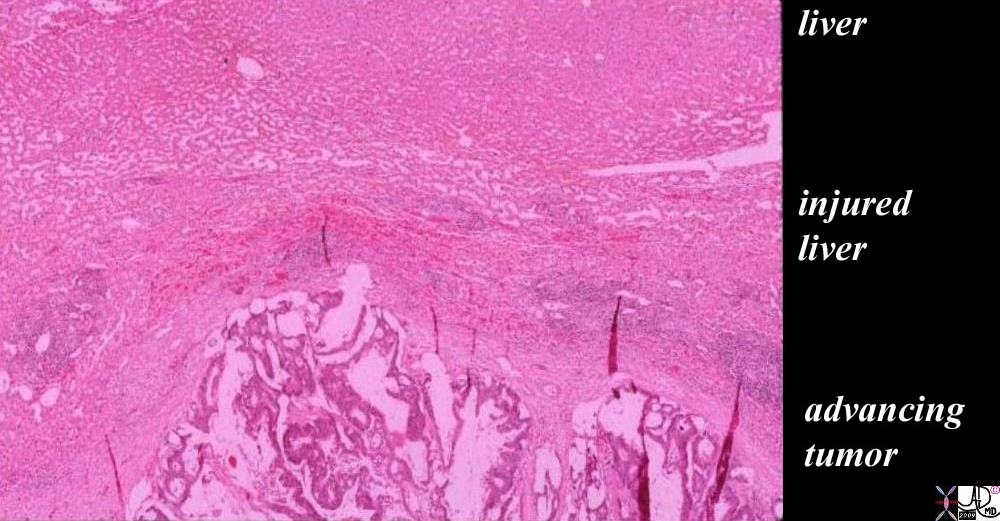
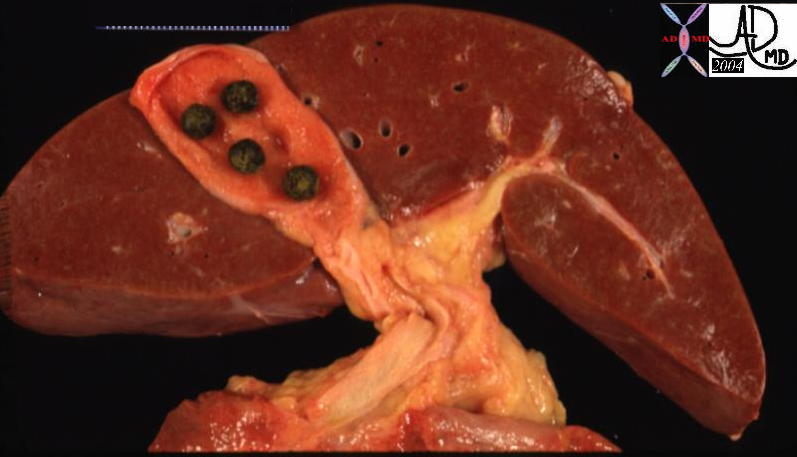
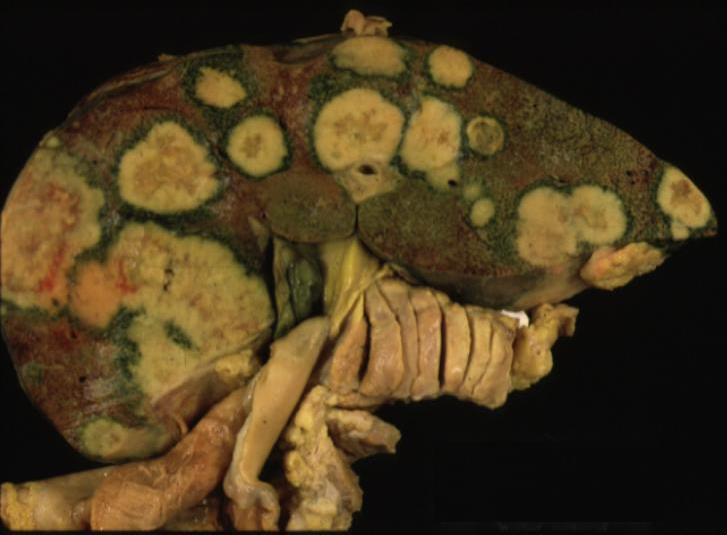
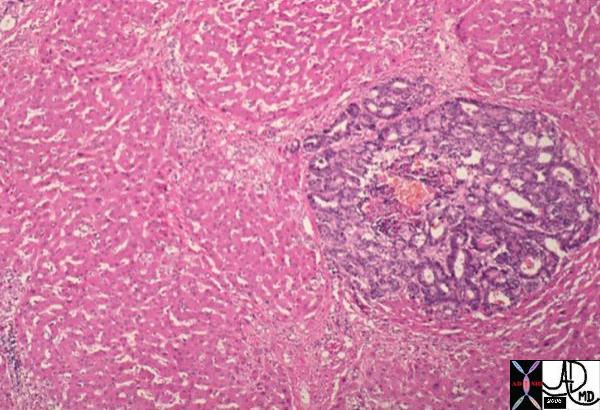 Normal Liver and Single Group of Cancer Cells
Normal Liver and Single Group of Cancer Cells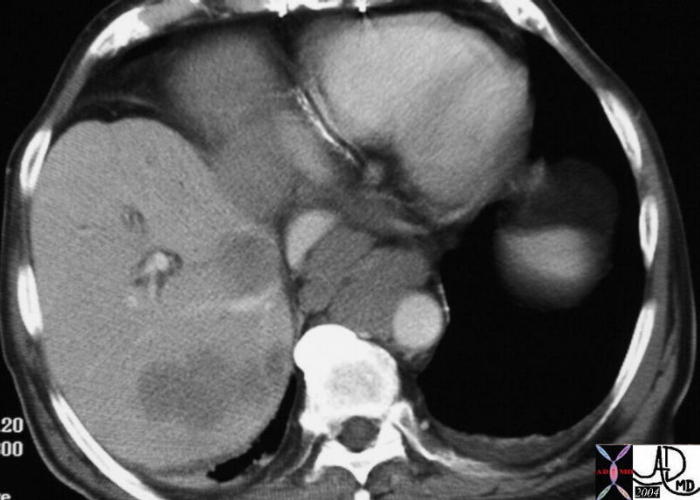
 Breast Carcinoma
Breast Carcinoma
 Components of the Breast
Components of the Breast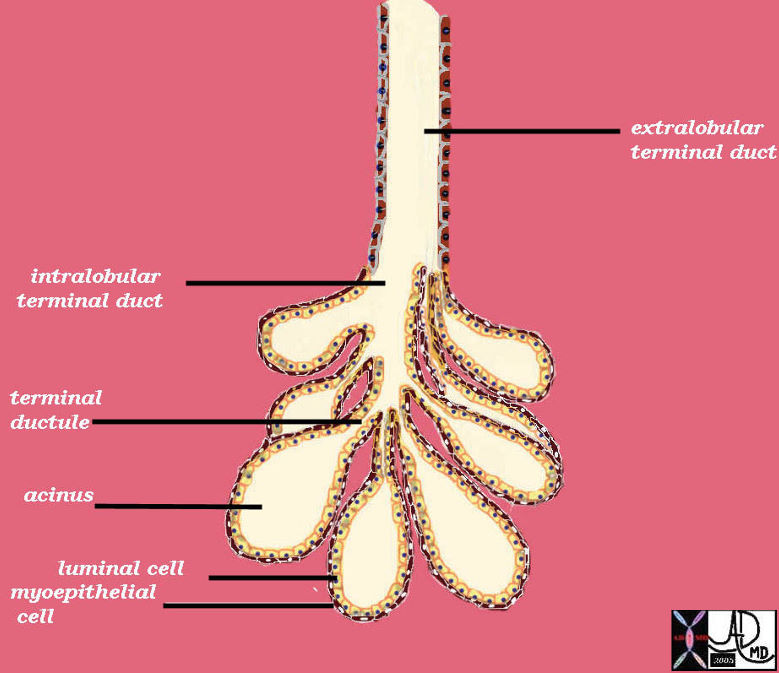
 Microscopic Histology of the Lobule
Microscopic Histology of the Lobule Spiculated Carcinoma
Spiculated Carcinoma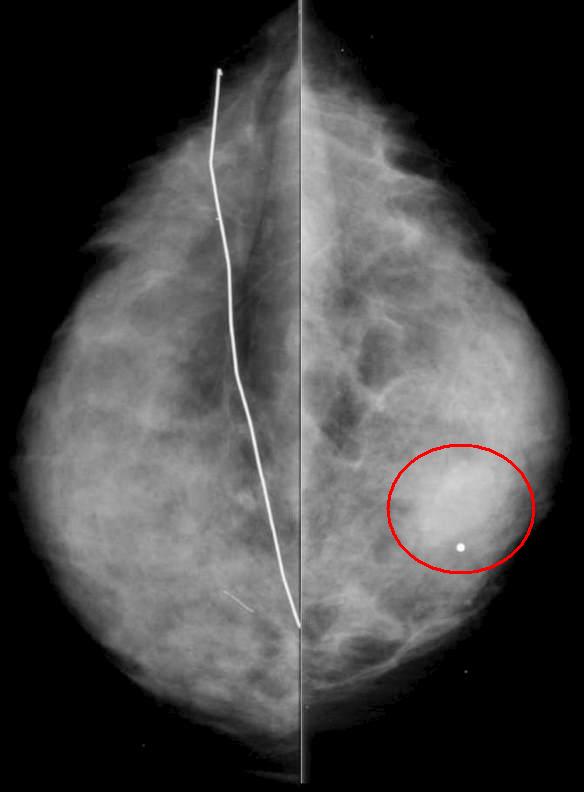 Soft Tissue Mass – Invasive Ductal Carcinoma
Soft Tissue Mass – Invasive Ductal Carcinoma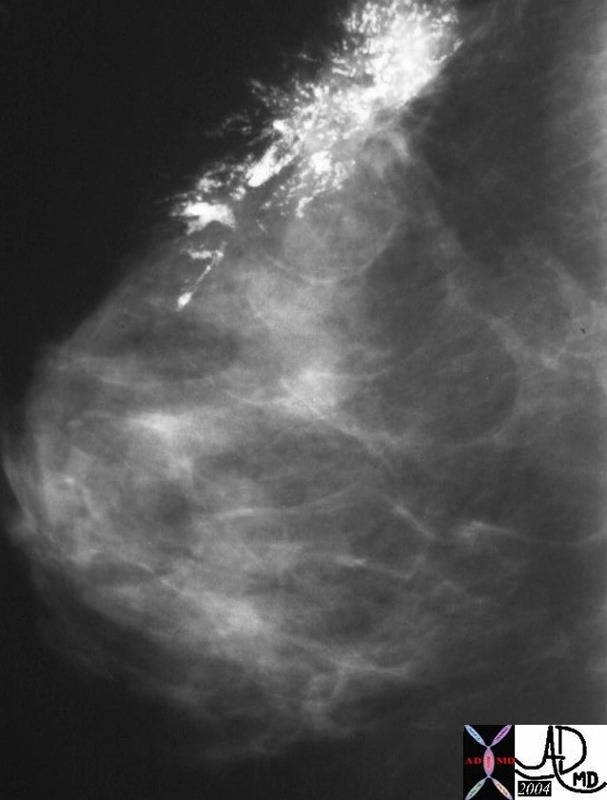 Invasive Ductal Carcinoma DCIS , Positive Nodes
Invasive Ductal Carcinoma DCIS , Positive Nodes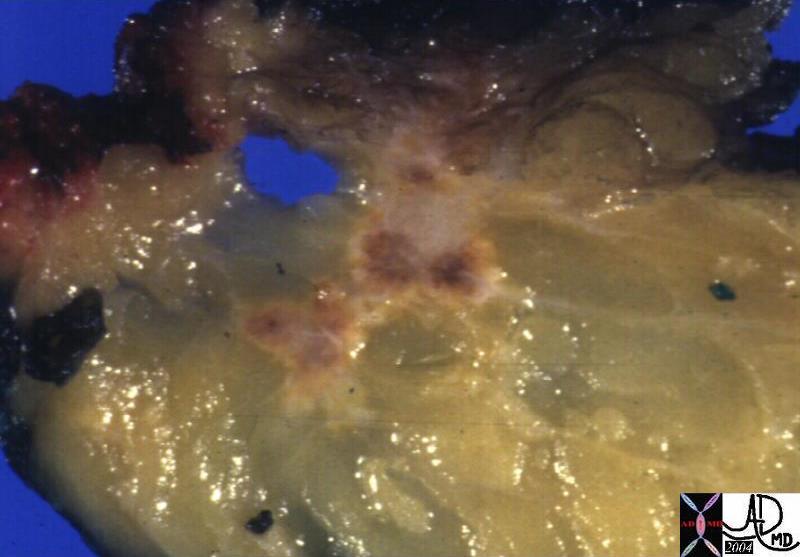 Invasive Scirrhous Ductal Carcinoma
Invasive Scirrhous Ductal Carcinoma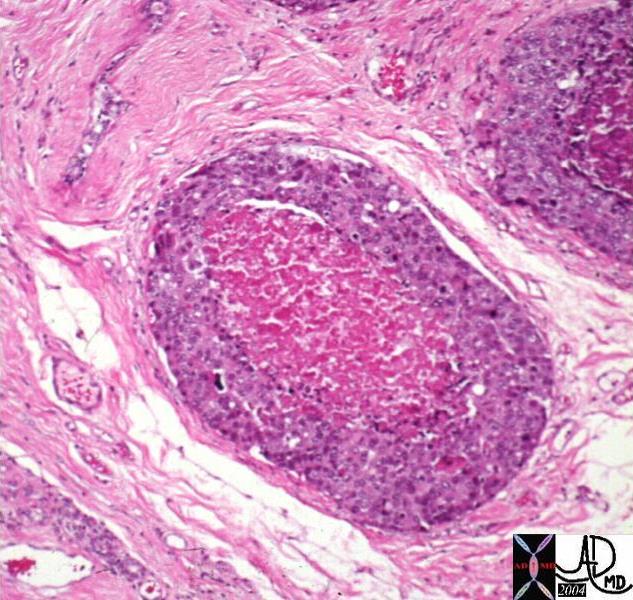 Infiltrating Ductal CArcinoma
Infiltrating Ductal CArcinoma Invasive Lobular Carcinoma
Invasive Lobular Carcinoma MRI – Invasive Lobular Carcinoma
MRI – Invasive Lobular Carcinoma Ill defined Mass in the Surical Specimen
Ill defined Mass in the Surical Specimen
 Histopathology of Invasive Lobular Carcinoma
Histopathology of Invasive Lobular Carcinoma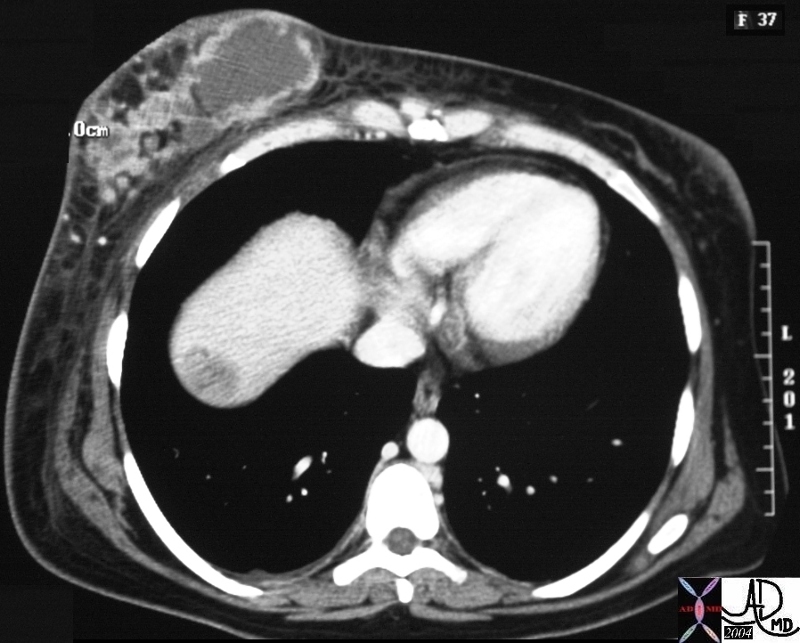 CT scan – Inflammatory Carcinoma
CT scan – Inflammatory Carcinoma Well Circumscribed Mass – Phyllodes Tumor
Well Circumscribed Mass – Phyllodes Tumor Magnified View of the Well Circumscribed Phyllodes Tumor
Magnified View of the Well Circumscribed Phyllodes Tumor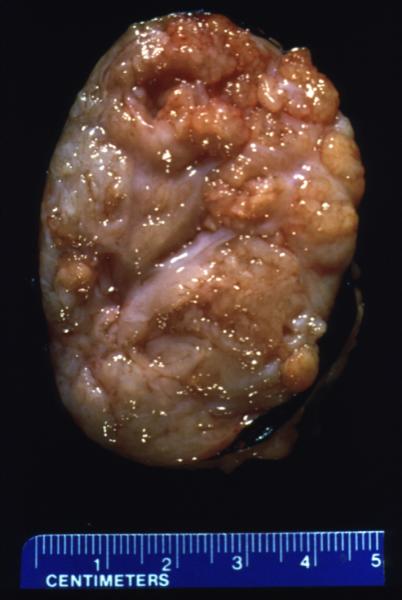
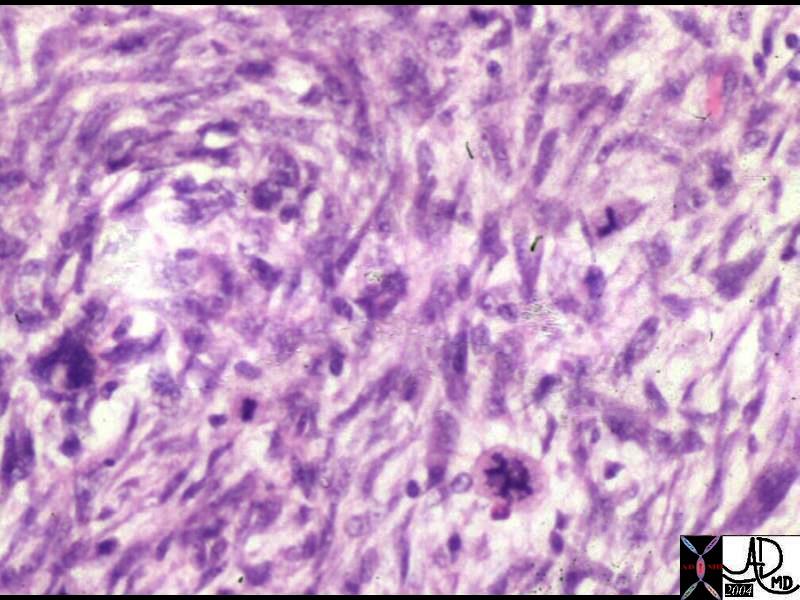 Histopathology of the Phyllodes Tumor
Histopathology of the Phyllodes Tumor Well Defined Mass
Well Defined Mass Ultrasound Showing a Cyst
Ultrasound Showing a Cyst Ultrasound Showing a Solid Lesion – Fibroadenoma
Ultrasound Showing a Solid Lesion – Fibroadenoma
 Microcalcifications Identified on Mammography
Microcalcifications Identified on Mammography Specimen Biopsy Confirms That Correct Tissue has Been Biopsied
Specimen Biopsy Confirms That Correct Tissue has Been Biopsied
 Cytopathology Specimen Infiltrating Ductal Carcinoma
Cytopathology Specimen Infiltrating Ductal Carcinoma Lymphatic supply of the breast
Lymphatic supply of the breast
 Lymph nodes
Lymph nodes